Chapter Five Review Sheet
advertisement

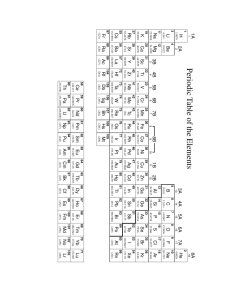
Review Sheet for Common Assessment 12 weeks- Includes Chapters 1 through 5 in the book 1. What element is located at 3d2? What family does it belong to? Titanium , transition metal 2. What are the semi-metals or metalloids on the periodic table? B, Si, Ge, As, Sb, Te and At 3. What family on the periodic table only has one valence electron? Alkali Metal Family 4. If an element has more than four valence electrons it is most likely a metal or a nonmetal? nonmetal 5. How many valence electrons are found in a calcium atom? 2 6. How many valence electrons are found in a Tungsten atom? 2 7. Which is more reactive, sodium or barium? Sodium 8. Which is more reactive, sulfur or chlorine? Chlorine 9. Which element would have the smaller radii, Chromium or potassium? Chromium 10. Which would have the higher electronegativity value, Lithium or Nitrogen? Nitrogen 11. Which of the following pairs belong to the same period? a) Na & Cl b) Na & Li c) Na & Cu d) Na & Ne a) Na & Cl 12. Define the following terms: Ionization energy: the amount of energy required to remove one electron from an atom Electronegativity: the ability of an atom to attract an electron in a chemical bond 13. Give the electron configuration for an excited Sulfur atom. 1s2 2s2 2p6 3s2 3p3 4s1 14. How many valence electrons does the element 5d6 have? 2 15. Give the electron configuration for O 2- . 1s2 2s2 2p6 16. Give the electron configuration for Na + . 1s2 2s2 2p6 17. Why does magnesium want to lose two electrons? If it loses 2 electrons then it will have the electron configuration of a noble gas 18. How does the shielding effect relate to a metal’s reactivity? Metals want a greater shielding effect because they can easily lose electrons, while nonmetals want less of a shielding effect so they gain electrons 19. Why does Fluorine have a higher ionization energy than Francium? The shielding effect is less for Fluorine, which means the valence electrons are closer to the nucleus thus needing more energy to remove one 20. Why does Fluorine have a higher electronegativity value than Francium? An electron can get closer to the nucleus of fluorine 21. What family is the most reactive metal family on the chart? Alkali metals 22. What family is the most reactive nonmetal family on the chart? Halogens 23. How many valence electrons does chlorine have? 7 24. How many electrons does chlorine need to gain or lose to become stable? Needs to gain 1 electron. 25. Who would have a larger atomic radii Calcium or Barium ? Barium 26. The speed of light is 3.0 x 10 8 m/s. If a star is 5.40 x 10 12 miles away, how many days would it take the light to reach earth? ( 1 km = .621 miles) 336 days 27. What is the difference between an isotope and an ion? An isotope has the same number of protons, but the number of neutrons have changed thus changing the atomic mass. An ion has the same number of protons, however electrons have either been lost ( positive ion) or gained ( negative ion) . 28. How many hours would it take to go 566 km, given you are traveling at 75 mph? ( 1 km = 0.621 mi) 4.69 hours 29. How much would a rock weigh if its density is 3.22 g/ml and its volume was 456 mL? 1470 g 30. What is the volume of a piece of aluminum given its density is 2.70 g/cm3 and its mass is 4.00 g? 1.48 cm3 31. 7.88 g X 4.5 mg = ______________ g or _________________ mg .035 g or 35,000 mg 32. Identify which property is chemical or intensive physical or extensive physical a) Ability to react with water (Chemical) b) Density ( Intensive) c) Color (Intensive) d) Rust (Chemical) e) Malleable(Intensive) f) Mass (Extensive) g) Length (Extensive) 33. Which of the following is a physical (P) or chemical change (C)? a) Burning (c) b) Melting (P) c) Evaporation (P) d) Breaking (P) e) Tarnish (c)