threat of domestic power generators to terrestrial ecosystem as
advertisement

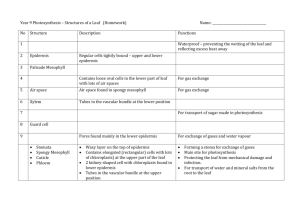
THREAT OF DOMESTIC POWER GENERATORS TO TERRESTRIAL ECOSYSTEM AS EXPRESSED IN SOME PLANT SPECIES. J. E. Otoide1 and J. Kayode2 Department of Plant Science, Ekiti State University, P.M.B 5363, Ado-Ekiti, Nigeria, 1 E-mail: joeotoidejo@yahoo.com, 2E-mail: jokayode@ymail.com Polluted populations of Euphorbia heterophylla, Chromolaena odorata Commelina diffusa and Kyllinga pumila were collected fresh from within 0.1-0.25m radii of the exhausted-pipe of power generators used for domestic purposes while their non-polluted counterparts were collected from distances of 100-110m away. The length and width of ten leaves, each of both polluted and non-polluted populations, were measured. Their average leaf areas (LA) were 2.60 + 0.1cm2, 14.16 + 0.4cm2, 5.19 + 0.8cm2 and 1.80 + 0.8 cm2 for the polluted populations of each of the species respectively while the average leaf areas (LA) of their non polluted counterparts were 4.18 + 0.7 cm2, 34.39 + 0.2cm2, 7.51 + 0.1cm2 and 10.76 + 0.3cm2 respectively. Damages such as plugged stomatal pores, epidermal cell aberrations and erosion, ruptured stomatal ledges, occasional leaf perforations, irregularly fused cell boundaries and glued leaf surfaces were noticeable in the leaves of the polluted populations. Such were not observed in the non-polluted populations. It was suggested that foliar morphology of these group of plants could serve as phytometer to gauge the threats of power generators used in homes to terrestrial ecosystem. Key words: Domestic power generator, terrestrial environment, plant species Introduction The ever increasing human desire for comfort and good living standard as well as industrial activities has warranted the use of cars, vehicles, power generating plants and other mechanical and electrical appliances (AFF, 1953). All of these are capable of generating phytotoxic air pollutants such as black carbon (carbon monoxide- CO), Carbon dioxide (CO2), Sulphur dioxide (S02), Nitrogen Oxides (NOx) and Peroxyacetylnitrates (PANs) (Bonnie and Joel, 2000; Socha, 2002). These harmful gases are emitted into the atmosphere where they pollute the air, and as a result of gravity, they are deposited on any living or non-living materials on the ground either in powdery state or in solution state (Lenntech, 2005). Air pollution is commonly experienced in major places such as busy roads with high vehicular traffic, homes especially from emissions by domestic power generators and industrial areas. Air pollutants in these places are capable of affecting plants by causing damages to the cuticular and subcuticular regions of the leaves. These damages could lead to the development of diseases and other problems on the affected plants (Pal et al; 2002 and Heather, 2003). Though the ecosystem has the capability to adjust to change and maintain equilibrium yet, if anyone of the ecosystem components is substantially altered due to natural or man made stress factors, the entire ecosystem can be affected (Adela, 1990). Previous studies had revealed that many flowers and vegetable crops suffered ill effects from air pollution caused by exhaust gases. Trees have also been killed by pollution. Air pollution causes rubber tires on automobiles to crack and become porous. Fine buildings become shabby, their walls blackened with soot as a result of the pollution that has settled on building stones and surface for years. This study presents the various cell alterations caused by gaseous pollutants on leaves of plant species growing near the exhaust-pipe of power generators used in homes. Materials and Methods Collection of Plant Samples Matured leaf samples of species of Euphorbia hererophylla, Chromolaena odorata, Commelina difusa and Kyllinga pumila were collected from within 0.1 - 0.25m radii of the exhaust–pipes of power generators used for domestic purposes especially by the elites in Benin City, Edo State, Nigeria. Another collection of same species were made in the same environment, but at distances of 100-110m away from the power generators. The first set of collections represented the polluted populations, while the second set represented the non-polluted populations which served as control for this study. Leaf Dimension Ten (10) leaves per species from the polluted and non-polluted areas were randomly selected, their sizes were measured with the aid of plastic ruler and the data were recorded. The Leaf Area (LA) of each leaf was then determined according to Duncan and Hesketh (1968), Remison and Lucas (1982) as: LA: L x W x 0.75 Where, L = Length of Leaf W = Width of Leaf 0.75 = Constant Preparation of Slides The epidermal peels of each leaf sample were obtained using the methods of Metcalfe and Chalk (1988) and Olowokudejo (1990). The leaves were placed, with the outer surface facing downward, on a flat surface and flooded with 8% sodium hypochlorite solution (NaOCl). An area of about 1cm square was removed from a central / standard position, always midway between the base and the apex of the leaves. The peels were mounted temporarily on slides. 10 slides (each of adaxial and abaxial surfaces) were prepared per population. Measurement of pores, guard cells and collection of data The slides were examined under the light microscope using x20 and x40 objectives. Data were collected from 10 microscopic fields selected at random from each slide. The length and width of stomatal pores and guard cells were measured using ocular micrometer. Data were collected from 25 stomata per leaf surface. This was done in 10 replications. Line drawings of the epidermal cells, stomata and subsidiary cells were made at different microscopic objectives. The data obtained were subjected to relevant statistics using mean, standard deviation and ANOVA. Significant differences were determined at p < 0.05. Results and Discussion The average leaf areas (cm2) of the plant samples from polluted and non-polluted sites were shown in Table 1 while their leaf epidermal features are summarized in Table 2. Illustrations of the epidermal features were shown in Figures1-4. The average leaf areas for the polluted and non-polluted populations of Euphorbia heterophylla were 2.60 + 0.1 and 4.18 + 0.7 respectively, while the average leaf areas for the pollution and non-polluted populations of Chromolaena odorata were 14.16 + 0.4 and 34.39 + 0.2 respectively. Similarly, average leaf areas of 5.19 + 0.8 and 7.51 + 0.1 were calculated for the polluted and non-polluted populations of Commelina diffusa respectively. Whereas, Kyllinga pumila had average leaf areas of 1.80 + 0.8 and 10.76 + 0.3 for the polluted and non-polluted populations respectively. Stomata were not observed in the upper epidermis of leaves of Euphorbia heterophylla and Kyllinga pumila. However, stomata were observed in the lower epidermis (Figs. 1 and 4). Chromolaena odorata and Commelina diffusa showed presence of stomata in the adaxial and abaxial surfaces of the leaves (Figs. 2 and 3). The stomatal pores in leaves of all the polluted plant populations were plugged by soot which hindered their measurements (Figs. 1a, 2a&b, 3a&c, 4a&c). On the other hand, the stomatal pores in leaves of the non-polluted plant populations were open and measured 2.01 + 0.16µm and 1.02 + 0.10µm as mean length and width of pores for Euphorbia heterophylla, while 1.92 + 0.71µm and 0.91 + 0.01µm were the mean length and width of Kyllinga pumila respectively. In Chromolaena odorata, the mean length and width of stomatal pores in the adaxial leaf surface of the non-polluted population were 1.99 + 0.82µm and 0.91 + 0.11µm, whereas, in the abaxial leaf surface, the mean length and width were 1.47 + 0.20µm and 0.70 + 0.40µm respectively. Also, the mean length and width of stomatal pores in leaves of the non-polluted populations of Commelina diffusa were, at the adaxial surface, 3.56 + 0.10µm and 0.80 + 0.01µm, while, at the abaxial surface, they were 2.56 + 0.13µm and 0.35 + 0.15µm respectively. The mean length and width of guard cells in the abaxial leaf surface of the polluted populations of Euphorbia heterophylla were 2.88 + 0.12µm and 1.62 + 0.4µm whereas; the non-polluted populations had 2.62 + 0.14µm and 1.59 + 0.16µm respectively. In the abaxial leaf surface of the polluted populations of Kyllinga pumila on the other hand, the mean length and width of guard cells were 2.44 + 0.41µm and 1.47 + 0.69µm while, in the non-polluted populations, the guard cells measured 2.91 + 0.05µm and 2.09 + 0.10µm respectively. Similarly, the mean length and width of guard cells in the adaxial leaf surface of the polluted populations of Chromolaena odorata were 2.84 + 0.25µm and 1.74 + 0.21µm while in the abaxial leaf surface, they were 2.02 + 0.36µm and 1.47 + 0.24µm respectively. The non-polluted populations of this species on the other hand, had guard cells which measured, at the adaxial leaf surface, 2.86 + 0.51µm and 1.47 + 0.92µm as mean length and width respectively but in the abaxial leaf surface, the mean length and width of guard cells were 2.81 + 0.01µm and 1.76 + 0.54µm respectively. The mean length and width of guard cells in the adaxial leaf surface of the polluted populations of Commelina diffusa were 3.62 + 0.10µm and 1.74 + 0.11µm while in the abaxial leaf surface they were 3.46 + 0.14µm and 1.72 + 0.21µm respectively. In the non- polluted populations of this species on the other hand, the mean length and width of guard cells in the adaxial leaf surface were 4.15 + 0.14µm and 2.17 + 0.05µm, whereas, in the abaxial leaf surface they were 4.06 + 0.50µm, and 2.88 + 0.08µm respectively. The nature of the epidermal cell walls in leaves of both populations of Euphorbia heterophylla was straight in the adaxial leaf surfaces but sinuous in the abaxial surfaces (Fig.1). In both surfaces of the two populations of Kyllinga pumila, the epidermal cells walls were straight but in the adaxial epidermis of Chromolaena odorata, they were slightly sinuous in the two populations but sinuous in the abaxial surfaces of both populations. Also, the epidermal cell walls in the adaxial and abaxial leaf surfaces of the polluted populations of Commelina diffusa were straight but were straight and sinuous in the adaxial and abaxial surfaces in the non-polluted populations. Deposits of soot particles were observed in most of the epidermal cells, guard cells and cell walls in the adaxial and abaxial leaf surfaces of the polluted populations of the species. The soot particles tend to obscure and damage most of the epidermal features in the leaf surfaces as they glued both surfaces of the leaves. Occasional perforation and eroded epicuticular wax were other observed damages on the leaves of the polluted populations. Conversely, the soot particles were not observed in the epidermal cells of the non-polluted populations of same plant samples and their leaves appeared undamaged. TABLE 1: Average Leaf Area (cm2) of plants from polluted and non-polluted populations S/No Species Populations* Polluted Non-polluted 1. Euphorbia heterophylla 2.60+0.1a 4.18+0.7b 2. Chromolaena odorata 14.16+0.4a 34.39+0.2b 3. Commelina diffusa 5.19+0.8a 7.15+0.1a 4. Kyllinga pumila 1.80+0.8a 10.76+0.3b * Figures with same letter in each row are not significantly different at 0.05 level of significance. TABLE 2: Leaf epidermal characteristics of the plant species from polluted and non-polluted populations. Descriptions Surface Mean length of pore (µm) Mean width of pore(µm) Mean length of guard cell (µm) Mean width of cell(µm) Nature of epidermal cell wall U L A B U Euphorbia A - heterophylla B - Chromolaena A Plugged odorata B 1.99+0.82 Commelina A Plugged diiffusa B 3.56+0.10 Kyllinga A - pumila B - L U Plugged - 2.01+0.16 - Plugged Plugged 1.47+0.20 0.91+0.11 Plugged Plugged 2.56+0.13 0.80+0.01 Plugged - 1.92+0.71 - L U Plugged - 1.02+0.10 - Plugged 2.84+0.25 0.70+040 2.86+0.51 Plugged 3.62+0.10 0.35+0.15 4.15+0.14 Plugged - 0.91+0.01 - L U 2.88+0.12 - 2.62+0.14 - 2.02+0.36 1.74+0.21 2.81+0.01 1.47+0.92 3.46+0.14 1.74+.011 4.06+0.50 2.17+0.55 2.44+0.41 - 2.91+05 - L U 1.62+0.41 Straight 1.59+0.16 Straight 1.47+0.24 Slightly Sinuous 176+0.54 Slightly Sinuous 1.72+0.21 Straight 1.47+0.69 Straight 2.09+0.10 Straight L Sinuous Sinuous Sinuous Sinuous Straight 2.88+0.08 Straight, Sinuous around the veins Anticlinal: Straight Periclinal: Sinuous Straight Straight = = = = Upper epidermis Lower epidermis Polluted Populations Non-Polluted Populations. Fig.1: (a) The abaxial epidermis of Euphorbia heterophylla from polluted area in Benin City, Nigeria x400. (b) The abaxial epidermis of E.heterophylla from non-polluted area in Benin City x400. (c)The adaxial epidermis of E. heterophylla from area polluted area in Benin City.x200. (d) The adaxial epidermis of E.heterophylla from non-polluted area in Benin City.x400 Fig.2: (a) The abaxial epidermis of Chromolaena odorata from polluted area in BeninCity x400. (b) The abaxial epidermis of C. odorata from non-polluted area in Benin City x400. (c) The adaxial epidermis of C.odorata from polluted area in Benin City x400 (d) The adaxial epidermis of C. odorata from non-polluted area in Benin City. x400. Fig.3: (a) The abaxial epidermis of Commelina diffusa from polluted area of Benin City x200. (b) The abaxial epidermis of C. diffusa from non-polluted area in Benin City x400. (c) The adaxial epidermis of C. diffusa from polluted area in Benin City x200. (d) The adaxial epidermis of C. diffusa from non-polluted area in Benin City x400. Fig. 4: (a) The abaxial epidemis of Kyllinga pumila from polluted area in Benin City x200. (b) The abaxial epidermis of K. pumila from non-polluted area in Benin City x400. (c) The adaxial epidermis of K. pumila from polluted area in Benin City x400. (d) The adaxial epidermis of K. pumila from non-polluted area in Benin City. x400. Plant samples collected within 0.1-0.25m from the exhaust-pipe of power generators used in residential area showed reduced leaf size, plugged stomatal apertures blurred epidermal surfaces and obscured cells (Tables 1-2 and Figs.1-4) These alterations might be attributed to the activities of soot particles (Black carbon) and other gaseous pollutants from the incomplete combustion of fossil fuel which the plant species were continuously exposed to, since these features were not observed in the non-polluted populations of the same species. These observations tend to confirm the previous assertions by Heather (2003) and Mandal and Mukherjee (2001) that harmful gaseous air pollution cause reduction in leaf size, plugged stomatal pores and injure plants leaves. Domestic power generators being stationary sources of toxic air pollutants emit harmful gases including soot particles (Black Carbon) directly onto any object located very close to their exhaust-pipes. During the field sampling, soot particles were found covering the leaf blades and parts of the stems of affected plants. Consequently, the affected portions appeared black. It is suspected that the black soot particles on the samples would have some negative ecophysiological effects, such as the screening out of solar energy (sunlight) and prevention of carbon dioxide assimilation in them. These would hinder photosynthetic, respiratory and transpiratory activities in the plants. This assertion is further buttressed by the fact that these plants possessed leaves that were poorly developed and had reduced leaf sizes. Whereas, similar plant species sampled from the non-polluted areas, showed proper leaf development with large leaf sizes (Tables1-2). This observation tends to support the previous assertions of Adela (1990) that air pollutants injure plants by damaging their foliage and impairing the process of photosynthesis (Food making). It further supports the assertions of Garner (2002) that high concentration of sulphur dioxide, nitrogen oxides and ozone in the atmosphere can affect plant growth and reproduction. They are capable of inhibiting photosynthesis carbon (sugar) production, alter carbon allocation to roots and stems, reduce carbohydrate formation of mycorrhizae, uptake of important minerals, root and stem growths. Results from this study (Tables 1-2) revealed that the polluted leaves were smaller in area than the non-polluted leaves in all the species sampled. The small nature of the leaves might debar them from capturing enough amount of light as the larger non-polluted leaves will do. It is further suspected that the deposits of soot particles and gaseous toxins in the leaf epidermis in the polluted population could initiate chemical reactions with moisture and to produce complex acids. This is pertinent, since Adela (1990) had earlier asserted that SO2 and Nitrogen oxides may enter leaves and then react with cell moisture to form complex acids. From the results, it is suspected that the damages and aberrations on the epidermis (Figs 1-4) of leaves in the polluted populations were caused by the gaseous air pollutants they were subjected to, since the non-polluted population did not show such damages and aberrations in their leaves. This further support the reports of Edwardo (2002) that air pollutants bind to plasma membranes, alter metabolism, destroy stomatal apertures, damage chloroplast thylakoid membranes,erode epicuticular wax, destroy epidermal cells and inhibit photosynthesis. In conclusion, the leaves of the plant species used for this study had expressed the threat of domestic power generators which they were exposed to, by showing various forms of damages and aberrations reported in this investigation. It could, therefore, be inferred that damages and aberrations resulting from gaseous pollutants will only be experienced by plant species growing at 0.1-025m to the exhaust-pipe of power generators and not those growing at 100-110m away from power generators. It is believed that the damages and aberrations which are the major threats exhibited by the species investigated in this study could serve as base line information of what would happen when species are exposed to air pollutants from power generators while the plant species could serve as phytometer to gauge the threats of power generators used in homes to terrestrial ecosystem which basically has plant species as its major components. It is, therefore, recommended that the Governments of Edo State and Nigeria in general should improve on electricity supply so as to reduce the rate at which her citizens use alternative source of power supply especially generators as they are capable of emitting harmful gaseous substances into the environment. Urban agriculture should be discouraged in residential areas where power generators are used for domestic purposes. Crops growing in such areas are liable to black soot particulates which could be deposited on their leaf surfaces. This will hinder their photosynthetic activities and consequently affect other physiological activities of the crops. If on the other hand urban agriculture is a necessity, the crops should be at least 100m away from the location of domestic power generators in the environs. References Adela B 1990 Acid Rain, Air Pollution and Forest Decline. CRS Report from Congress. 86031. http://www.ncsonline.org/nce/crsreports/natural/nrgen-15.cfm. AFF 1953 Aviation and the Global Environment, Intergovernmental Panel on Climate Change, IPCC Special Report, Geneva. Bonnie A, Joel K 2000 Trees for Problem Landscape Sites-AirPollution. Virginia Cooperative Extension. Pub.No 430022. Duncan W G, Hesketh J D 1968 Net Photosynthetic rates, relative leaf growth rates, and leaf numbers of 22 races of Maize grown at eight temperatures. Crops Science 8 670-674. Edwardo Z 2002 The effect of air pollution on Plants. Back to Plant Phys. Net. Essay 25.1 University of California, Los Angeles, USA. Garner J H B 2002 Air Pollutants, Plant Response, Soil Microbes and Ecosystem Biodiversity. Environews 8 (3) 1-7. Heather G 2003 Effects of Air Pollution on Agricultural Crops. FACTSHEET ISSN 1198-712X. . Lenntech I 2005 Air Pollution. Frequently Asked Questions. Lenntech Water Treatment and Air Purification. Holding B.V. Rotterdamseweg 402m, 2629 H.H Deft, The Netherlands. Mandal M, Mukherjee S 2001 A study on the activities of a few tree radicals scavenging enzymes present in five road side plant. Journal of Environmental Biology 22 (4) 301-305 Metcalfe C R, Chalk L 1988 Anatomy of Dicotyledons.Clarendon Press, Oxford. Olowokudejo J D 1990 Comparative Morphology of leaf epidermis in the genus Annona (Annonanceae) in West Africa. Phytomorphology 40 407-422 Pal A, Kulshreshtha K, Ahmad K J, Behl H M 2002 Do leaf surface characters play a role in plant resistance to auto-exhaust pollution? http://www.ingentaconnect.com. Remison S U, Lucas E O 1982 Effects of planting density on leaf area and productivity of two maize cultivars in Nigeria. Experimental Agriculture 18 98 -100. Socha T 2002 Pollution Damage to plants: Air pollution causes and effects. http://heathandenergy.com/airpollutioncauses.htm.