Lecture 3A Scientific Math NOTE SHEET Temperature Conversion 8
advertisement

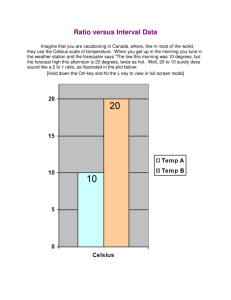
Chemistry Lecture 3A Scientific Math Name ___________________________________ 8-18-14 Over the next few lessons you will learn some basic math essential to studying chemistry: _________________________________ _________________________________ _________________________________ I CAN ______________________________________________________________________________ ______________________________________________________________________________ TEMPERATURE vs HEAT • • • • • What is __________________ and how is it different from _______? Temperature and Heat are ________ the same thing. How are they different? Remember ______________________? _____________ is a measure of the ____________________ possessed by a substance. ___________________ is a measure of the ______________ possessed by the particle of a substance. EXAMPLE • Two beakers of water are placed on a hot plate and heated until both reach the boiling point of water, 100 oC. • • • • What is the ______________of each beaker? _______ Which beaker of water has _________________________? The ___________________________________________. TEMPERATURE SCALES FAHRENHEIT SCALE Since the invention of the first modern thermometer in 1724, scientists have created a number of temperature scales. • • • • • Since the invention of the first modern thermometer in 1724, scientists have created a number of temperature scales. A common widely used scale is the ________________________ scale. Freezing Point (water) ___________. Boiling Point (water) ____________. _____________________________________________ between FP and BP. • Temperatures can be __________________________________________. CELSIUS TEMPERATURE SCALE • • • • • • • • The Celsius Scale is the preferred scale for most scientific work. Part of the __________________________________________. Freezing Point is ________________. Boiling Point is _________________. ___________________________________________________ between FP and BP. Temperatures can be _________________________________________________. For ____________ science work the __________________________ is fine. However, in some calculation, temperatures _______________________________ present a problem. Example Volume/Mass calculations cannot use temperatures less than zero because volume or masses _________________________________. In these instances, a third scale is used. KELVIN TEMPERATURE SCALE • • • • • • The ____________________ is based on the ___________ and ____________________________. For ___________________________ (+ or -) Kelvin, matter expands or contracts by________________ of its original volume. If matter’s temperature could be lowered to __________________, matter would have a _______________________, which is THEORETICALLY impossible. This temperature is known as _____________________________________. At this temperature matter would possess ______________________________________! Scientist have come within a few millionths of a degree of absolute zero! TEMPERATURE CALCULATIONS/CONVERSION BETWEEN SCALES Often in scientific work, it is necessary to convert temperatures from one system to another. This is easily done with the appropriate equation. CONVERTING A FAHRENHEIT TEMPERATURE TO CELSIUS • To convert a temperature given in oF to Celsius, use this equation: CONVERTING A CELSIUS TEMPERATURE TO FAHRENHEIT • To convert a temperature in oC to Fahrenheit, use this equation: CONVERTING CELSIUS TO KELVIN or KELVIN TO CELSIUS (Generally just 273 is fine!) CONVERTING FAHRENHEIT TO KELVIN • To convert Fahrenheit to Kelvin: [COMPLETE PRACTICE PROBLEMS]
![Temperature Notes [9/22/2015]](http://s3.studylib.net/store/data/006907012_1-3fc2d93efdacd086a05519765259a482-300x300.png)









