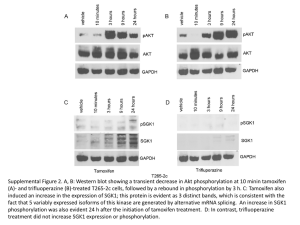
Schlosser/Timmer D.2. Quantitative Phosphoproteomics State of the
advertisement

Schlosser/Timmer D.2. Quantitative Phosphoproteomics State of the Art Reversible phosphorylation on Ser, Thr and Tyr residues is the most significant posttranslational modification in higher eukaryotes, which affects many cellular processes, such as protein-protein interaction, signalling, cell cycle, circadian rhythm, and many more. Mass spectrometry is more and more the method of choice for the analysis of protein phosphorylation and a large variety of different methods has been developed and optimized during the last decade (for review see [1]). In our lab we have established a platform that comprises a variety of different methods for quantitative phosphoproteomics. We have established an automated procedure for phosphopeptide enrichment using TiO2 nano columns [2]. Using a set of different proteases has proven to be a good strategy for improving the comprehensiveness of phosphorylation site mapping [2]. We use different fragmentation methods (collisioninduced dissociation (CID) as well as electron transfer dissociation (ETD)) for phosphopeptide analysis. This has proven to increase the number of identified phosphopeptides and to improve the localization of phosphorylation sites [3]. In addition we have developed a software tool that improves the identification of phosphopeptides and the pinpointing of phosphorylation sites compared the commercial search engine Mascot, especially for multi-phosphorylated peptides [4]. For exact relative quantitation of phosphorylation we have established different methods for metabolic labelling (SILAC and 15N-labeling). Taken together, phosphoproteomics is still a very challenging task and method development and optimization is still urgently needed. Thereby, the focus of our lab is on more targeted phosphoproteomics approaches. Work Plan Qualitative Phosphoproteomics In the first instance we will apply our established methods for in-depth mapping of phosphorylation sites of known key players of different signalling pathways, such as JAK1STAT3, SMAD2/3-SnoN, MAP kinase (ERK1/2), PI3 Kinase (Akt) (AG Klingmüller), NFkappaB (AG Bode) or betaCatenin. We will combine different methods, such as the multi-protease approach [2], phosphopeptide enrichment with TiO 2 [2], and ETD for obtaining the best possible coverage of phosphorylation sites. In parallel we will further optimize methods for the analysis of multi-phosphorylated peptides. Peptides with several phosphate residues are, due to a combination of different reasons, especially difficult to detect and current methods show a strong bias against multi-phosphorylated peptides. Quantitative Phosphoproteomics In the next step we will establish robust methods for exact relative quantitation of protein phosphorylation. Since primary hepatocytes are mainly used in the different projects, strategies for metabolic labeling, such as SILAC are hardly applicable. Therefore we will focus on the optimization of quantitation using Multiple Reaction Monitoring (MRM) on a triple quadrupole instrument [5] applying either label-free strategies [6] or strategies using synthetic internal standard peptides labeled with stable heavy isotopes ( 13C or 15N). As an alternative strategy we will evaluate the use of metabolically stable isotope-labeled phosphoproteins as internal standards (culture-derived isotope tags (CDITs)) [7]. These proteins can be obtained cell culture. Besides relative quantitation of phosphorylation (i.e. phosphorylation at S-338 has increased by 50%), quantitation of the phosphorylation stoichiometry (i.e. phosphorylation stoichiometry at S-338 has changed from 40% phosphorylated to 60% phosphorylated) is often important for modeling biological processes. Therefore, we will also establish robust methods that allow for the determination of the phosphorylation stoichiometry [6]. Our aim for the first three years is to establish a robust platform that allows highly sensitive, accurate and comprehensive quantitation of protein phosphorylation. This platform will be open to all projects of the HepatoSys program. Finally, we want to develop methods that allow for the analysis of fast phosphorylation/dephosphorylation processes [8]. Literature [1] Nita-Lazar A, Saito-Benz H, White FM (2008) Quantitative phosphoproteomics by mass spectrometry: past, present, and future. Proteomics 8:4433-4443. [2] Schlosser A, Vanselow JT, Kramer A (2005) Mapping of phosphorylation sites by a multi-protease approach with specific phosphopeptide enrichment and nanoLC-MS/MS analysis. Anal. Chem. 77: 5243-5250. [3] Molina H, Horn DM, Tang N, Mathivanan S, Pandey A (2007) Global proteomic profiling of phosphopeptides using electron transfer dissociation tandem mass spectrometry. Proc. Natl. Acad. Sci. 104:2199-2204. [4] Schlosser A, Vanselow JT, Kramer (2007) A Comprehensive phosphorylation site analysis of individual phosphoproteins applying scoring schemes for MS/MS data. Anal. Chem. 79: 7439-7449. [5] Wolf-Yadlin A, Hautaniemi S, Lauffenburger DA, White FM (2007) Multiple reaction monitoring for robust quantitative proteomic analysis of cellular signalling networks. Proc. Natl. Acad. Sci. 104:5860-5865. [6] Steen H, Jebanathirajah JA, Springer M, Kirschner MW (2005) Stable isotope-free relative and absolute quantitation of protein phosphorylation stoichiometry by MS. Proc. Natl. Acad. Sci. 102:3948-3953. [7] Ishihama Y, Sato T, Tabata T, Miyamoto N, Sagane K, Nagasu T, Oda Y (2005) Quantitative mouse brain proteomics using culture-derived isotope tags as internal standards. Nature Biotech. 23:617-621. [8] Dengjel J, Akimov V, Olsen JV, Bunkenborg J, Mann M, Blagoev B, Andersen JS (2007) Quantitative proteomic assessment of very early cellular signalling events. Nature Biotech. 25:566-568. Five major recent subject-related publications Maier B, Wendt S, Vanselow JT, Wallach T, Reischl S, Oehmke S, Schlosser A, Kramer A (2009) A large-scale functional RNAi screen reveals a role for CK2 in the mammalian circadian clock. Genes Dev 23:708-718. Schlosser A, Vanselow JT, Kramer (2007) A Comprehensive phosphorylation site analysis of individual phosphoproteins applying scoring schemes for MS/MS data. Anal. Chem. 79: 7439-7449. Vanselow K, Vanselow JT, Westermark PO, Reischl S, Maier B, Korte T, Herrmann A, Herzel H, Schlosser A, Kramer A. (2006). Differential effects of PER2 phosphorylation: molecular basis for the human familial advanced sleep phase syndrome (FASPS). Genes Dev 20:2660-72. Schlosser A, Amanchy R, Otto H (2006) Identification of tyrosine-phosphorylation sites in the nuclear membrane protein Emerin. FEBS J. 273:3204-3215. Schlosser A, Vanselow JT, Kramer A (2005) Mapping of phosphorylation sites by a multiprotease approach with specific phosphopeptide enrichment and nanoLC-MS/MS analysis. Anal. Chem. 77: 5243-5250.