Applied Chemistry
advertisement

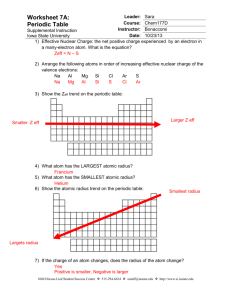
Academic Chemistry Section 8.1 Main Group Elements Name________________________________ Date__________________________ Study Guide From the following list of elements, select the one that best fits each of the descriptions below. aluminum boron calcium fluorine iodine magnesium oxygen nitrogen phosphorus potassium tin sulfur ____Oxygen_________ 1. The most abundant element on Earth. ____Iodine___________ 2. Needed for proper functioning of the thyroid gland. ___Potassium_________ 3. Reacts violently with water. _____Tin____________ 4. Used to make alloys such as bronze, solder, and pewter. ___Phosphorus_______ 5. A group 15 element used in making matches. ___Calcium__________ 6. An important element in bones and teeth. _____Sulfur__________ 7. Extracted from underground mines with hot water. _____Fluorine________ 8. Its compounds help prevent tooth decay. ____Magnesium_______ 9. Its atoms are found at the center of chlorophyll molecules. ____Aluminum_______ 10. The most abundant metallic element in Earth’s crust. _____Boron__________ 11. Used in the production of water softeners and certain types of glass. ____Nitrogen_________ 12. Obtained from liquid air by fractional distillation. Answer the following questions. 13. In general, how does atomic radius change across a period from left to right? It decreases 14. How does the size of a metallic atom change when it loses electrons to become a cation? It becomes smaller 15. How does the size of a metallic atom change when it gains electrons to become a anion? It becomes larger Compare each of the following pairs of elements, atoms, or ions by underlining the correct word in the pair. 16. Cesium is more/less active than sodium 17. Sodium is more/less active than magnesium 18. An aluminum ion has a larger/smaller radius than a magnesium ion. 19. An aluminum ion has a larger/smaller radius than a gallium ion. 20. At atom of fluorine has a larger/smaller radius than does a atom or iodine. 21. Sulfur is more/less active than selenium. 22. Antimony is more/less metallic than arsenic. 23. A halogen is more/less likely to occur in the free state than are members of the nitrogen family. 24. An atom of scandium is larger/smaller than an atom of titanium. Write or complete the chemical equations for each of the following chemical reactions: 25. Magnesium metal reacting with oxygen. 2 Mg + O2 2 MgO 26. Carbon reacting with oxygen. C + O2 CO2 27. NaF(s) Na+(aq) + F-(aq) 28. 2H2O2 2H2O + O2 29. 2KClO3 2KCl(s) + 3O2(g) 30. Ca5(PO4)3OH + F- Ca5(PO4)3F(s) + OH-(aq) 31. 4P(white)(s) + 5O2(g) P4O10(s) + heat