Ionic Compounds with Multivalent Metals Worksheet
advertisement

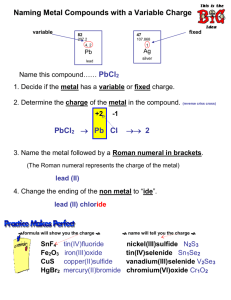
Sept 27/28 ` SNC2D - Chemistry Ionic Compounds with Multivalent Metals Most transition metals and some metals in group 14 (or IVA) are multivalent (they have more than one possible valence). They can form different numbers of bonds depending on the conditions. There are 2 systems of rules for naming compounds with metals of variable valence: IUPAC (international) and “Classical” (“ous-ic”). International System Writing Formulas: 1. Use the regular methods for writing formulas of ionic compounds (ionic charge or criss-cross). The metal is written first, and then the non-metal. 2. For the metal, use the charge given in brackets. Examples: copper (II) nitride Writing Names: tin (IV) oxide 1. From the formula, “back-cross” the subscripts – this will give you the valences. 2. Write the names of the elements in the same order as the formula (metal first, followed by the non-metal with “ide” suffix) 3. Use Roman numerals in brackets following the metal to indicate the charge. Examples: FeI2 SnO Classical System (“ous-ic”) Writing Formulas: 1. Use the regular methods for writing formulas of ionic compounds (ionic charge or cross-over). The metal is written first, and then the non-metal. 2. If the metal has the suffix “ous”, use the lower valence. If it has the suffix “ic”, use the higher valence. See the handout with the list of elements with variable valences Examples: Cuprous fluoride Writing Names: ferric sulfide 1. From the formula, “back-cross” the subscripts – this will give you the valences. 2. Write the names of the elements in the same order as the formula (metal first, followed by the non-metal with “ide” suffix) 3. For the metal name, use the Latin name, and add the suffix “ous” if the low valence was used, or the suffic “ic” if the high valence was used. Sept 27/28 Examples: ` Cu3N2 Practice I. Write the correct formulas for the following: SNC2D - Chemistry SnS 8. gold (I) sulfide 1. cobalt (II) sulfide 9. tin (IV) carbide 2. chromium (III) bromide 10. copper (II) nitride 3. lead (II) oxide 11. tin (IV) iodide 4. gold (I) oxide 12. copper (I) oxide 5. tin (II) sulfide 13. lead (III) phosphide 6. iron (III) nitride 14. Manganese (IV) oxide 7. bismuth (V) carbide 15. Nickel (III) sulfide Practice II. Write 2 names for the following, using IUPAC rules 1. SnF4 2. FeS 3. PbO2 4. HgCl 5. Ni2O3 6. Cu3N 7. AuF3 8. CuI 9. PbCl4 10. CuCl2 11. CoF3 12. FeO 13. MnCl7