Ch 6 & 7 Study Guide
advertisement

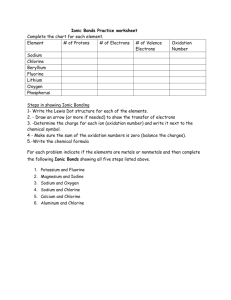
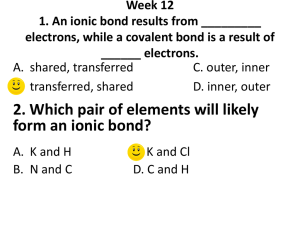
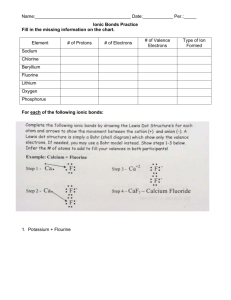
Name:___________________ Date:___________ Chapter 6 & 7 Test Study Guide 1. Identify each element as a metal, nonmetal, or metalloid. a. Iron b. Germanium c. Silicon d. Chlorine e. Magnesium f. Nitrogen 2. Identify each element as a solid, liquid, or gas a. Bromine b. Magnesium c. Nitrogen d. Krypton e. Copper 3. What are the properties of metals and nonmetals? 4. Who organized the elements into a periodic table in order of increasing atomic mass? 5. What are the specific names of the following groups? a. Group 1A b. Group 2A c. Group 7A d. Group 8A 6. Name an element that would have properties similar to Potassium (K). 7. What is an ion? Cation? Anion? 8. What is an ionic bond? 9. Classify each of the following as a cation or an anion. a. Na+ b. Ic. Ca2+ d. O2- 10. How many valence electrons are in the following atoms? e. Calcium f. Sulfur g. Silicon h. Chlorine 11. How many electrons are in the highest occupied energy level of atoms in Group 3A elements? 12. Draw the Lewis Dot Structures for the following atoms: a. Calcium b. Sulfur c. Silicon d. Chlorine 12. What is the octet rule? 13. What is the charge of the ions formed by elements in the following groups to achieve a noble-gas electron configuration? a. Group 1 b. Group 2 c. Group 6 d. Group 7 14. How many electrons does oxygen gain to achieve a noble-gas electron configuration? 15. What is the formula of the ion formed when nitrogen achieves a noblegas electron configuration? 16. Write the formula of the ionic compounds formed when the following elements combine. a. Sodium and Bromine b. Sodium and Sulfur c. Calcium and Iodine d. Aluminum and oxygen e. Barium and Chlorine 14. Ionic compounds are normally in which physical state at room temperature? 15. Why are metals electrical conductors? 16. What is an alloy? Why are they important?