*6th Grade Science-Chapter 5 Study Guide Lesson 5.1: Observing
advertisement

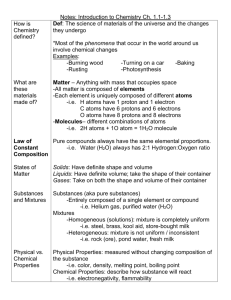
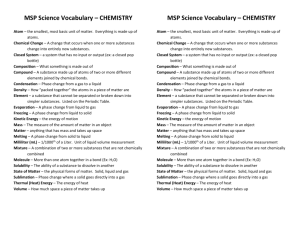
*6th Grade Science-Chapter 5 Study Guide Lesson 5.1: Observing Chemical Change *Changes in matter can be described in terms of physical changes and chemical changes. *Chemical reactions involve changes in properties and changes in energy that you can often observe. Physical change-any change that alters the form or appearance of a substance but does not change it into another substance. Ex: bending, cutting, freezing, and boiling are examples of physical changes. Chemical change-any change that produces one or more new substances. Chemical changes are caused by a chemical reaction. When a substance undergoes a chemical change, it results in different physical properties as well. Ex: burning and corrosion (rusting and tarnishing) are examples of chemical changes. Atoms form chemical bonds when they share or transfer electrons. Chemical changes occur when existing chemical bonds break and new bonds form. (Ex: 2 H2O + O2 2 H2O2 ) Reactants-substances that undergo chemical changes (the compounds on the left side of the arrow) Products-the newly formed substances that result from a chemical change (the compounds on the right side of the arrow) reactant + reactant product + product A precipitate is a solid that forms from liquids that undergo chemical changes in a chemical reaction. A gas can form from a solid or liquid as a result of chemical changes. A color change can occur as a result of chemical changes. Exothermic reaction- net energy is released from a chemical reaction (usually this energy is heat) Endothermic reaction- net energy is required for the chemical reaction to take place 5.2 Describing Chemical Reactions *A chemical equation tells you the substances you start with in a reaction and the substances that are formed at the end of the reaction. *In a chemical reaction, all of the atoms present at the start of the reaction are present at the end of the reaction. *Three types of chemical reaction are synthesis, decomposition, and replacement. Law of conservation of mass –states that during a chemical reaction, matter is not created or destroyed. Open system-matter can enter form or escape (Ex: open fish bowl) Closed system-matter does not enter of leave (Ex: a closed bottle) Balancing chemical equations: 1. write the equation 2. count the atoms on both sides of the equation 3. use coefficients to balance the number of atoms on each side Coefficients- a number placed in front of a chemical formula in an equation. Example Problem#1: hydrogen and oxygen combine to form water 1. H2 + O2 H2O 2. There are two atoms of hydrogen and two oxygen atoms on the left; there are two hydrogen atoms, but only one oxygen atom on the right 3. 2 H2 + O2 2 H2O is now balanced Example Problem #2: iron and oxygen react to form iron oxide 1. Fe + O2 FeO 2. There are two atoms of oxygen and one atom of iron on the left side; there is only on atom of iron and one oxygen atom on the right side. 3. 2 Fe + O2 2 FeO is now balanced Synthesis- reaction when two or more elements or compounds combine to form a more complex substance/product (Ex: A + B C) Decomposition-reaction when a compound breaks down into 2 or more simpler products (Ex: C A + B) Replacement reaction where one element replaces another element in a chemical reaction (Ex: AB + CD AD + CB) 5.3 Controlling Chemical Reactions *All chemical reactions need a certain amount of activation energy to get started Activation energy- the minimum amount of energy needed to start a chemical reaction *Factors that can affect rates of reactions include surface area, temperature, concentration, and the presence of catalysts and inhibitors. Concentration- the amount of a substance in a given volume Catalyst-increases the reaction rate by lowering the activation energy needed Enzyme- a type of biological catalyst (Ex: digestive enzymes in the human body) Inhibitor-a material used to decrease the rate of a chemical reaction (Ex: food preservatives) **In addition to this study guide and the vocabulary terms, please study the lab questions and textbook activities we did in class to help you prepare for the test.