Life on Earth Exists because of H2O
advertisement

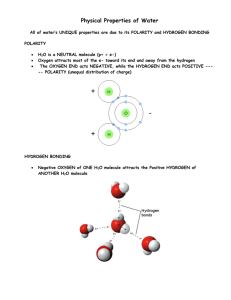
Life on Earth Exists because of H2O 1. Cells are mostly water 70-95%. 2. 3/4th of Earth’s surface is covered by water. 3. Water is a polar molecule: a.opposite ends of the molecule have opposite charges b.opposite charges attract the molecules to H-bond (each H2O molecules can form H-bonds with at least 4 others) c.this accounts for the properties of water 4. Properties of Water: a. Cohesion: Hydrogen bonds hold individual water molecules together it acts as one substance. (attraction that water has for itself) b. Adhesion: Clinging of water to another substance (the attraction that water has for other molecules) c. Surface Tension: (Is due to cohesion) it is a measure of the force necessary to stretch or break the surface of a liquid. d. Specific Heat: The amount of heat that must be absorbed or lost for 1g of that substance to change its temperature by 10C. 1. Heat is a measure of the total kinetic energy in a system. 2. Temperature measures the intensity of heat due to the average kinetic energy in molecules. a. Temperature is measured on the Celsius scale (Celsius = C) -H2O freezes at 00C -H2O boils at 1000C b. A calorie is a measure of heat. -One calorie (cal) is the amount of heat energy necessary to raise the temperature of one g of water by 10C - A kilocalorie (kcal) is the amount of heat energy necessary to raise the temperature of 1000 g of water by 10C -The specific heat of H2O is: 1cal per gram per degree Celsius: 1cal/g/0C -water has a high specific heat -water can absorb or release relatively large amounts of heat with only a slight change in its own temperature. -water can stabilize temperature 3. Water’s high specific heat is due to H-bonding a. heat is absorbed to break hydrogen bonds b. heat is released when H-bonds form e. Heat of Vaporization- the quantity of heat that a liquid must absorb for 1g of it to be converted from liquid to gas. -H2O has a high Heat of Vaporization due to (H-bonds which must be broken) -moderates environmental and body temp. 1. Evaporation: water molecule is energized, breaks H-bond, and turns into gas. Energy is taken from the system. a. convection cells-water absorbs heat in tropics through evaporation and releases it through condensation and precipitation Ex. – Tropics have moderate temp fluctuations, poles have extremes -Biomes - oceans same temp day & night vs. dessert with temperature extreme -Kendall vs. Mia Beach - sweating cools off animals - transpiration cools off plants f. Ice- water is less dense as a solid than as a liquid (at 40C, ice is 10% less dense than H2O liquid) 1. water expands as it solidifiesa. H-bonds do not have enough energy to break at 00C b. ice is a crystal lattice with each water molecule bonded to 4 others 2. Ice floats a. prevents ponds, lakes, oceans from freezing solid b. insulates g. Water is the ‘universal’ solvent of life 1. solution=homogeneous mixture composed of solute (subs, that is dissolved) and the solvent (the dissolving agent) a. aqueous solution = (water = solvent) 2. polarity of water allows it to form Hbonds with charged and polar mol. -ex. NaCl 3. hydrophilic substances have an affinity (form H-bonds) and/or dissolve in water. (ionic/polar subs.) hydrophobic substances do not have an affinity for water. (covalent/nonpolar/non-ionic subs.) ex. Lipids h. dissociation of water an H atom shared by two water molecules shifts from one molecule to the other. 1. Hydrogen ion – the Hydrogen atom leaves its electron behind and is transferred as a single proton (H+ ) 2. Hydroxide ion – the water molecule that lost the proton is an (OH-) 3. Hydronium ion – the water molecule with the extra proton is an ( H3O+) H2O ------- H+ + OH- 4. Hydrogen and hydroxide ions are very reactive and their concentrations can affect proteins and other molecules in a cell. 5. The pH scaledescribes how acidic or basic a solution is a. Acid – (proton donor) increases the H+concentration in a solution b. Base- ( proton acceptor) Reduces the H+ concentration c. in any solution the product of the H+ and OH- concentrations is constant at 10-14. [H+] [OH-] = 10-14 (1) in a neutral solution [H+] = 10-7 M [OH-] = 10-7 M (2) adding acid to a solution shifts the balance toward H+ conc. (3) adding base to a solution shifts the balance toward OH- conc. d. The pH scale is used to express the concentrations of Hydrogen and the Hydroxide ions. (1) neutral solution pH = 7 (2) Acidic solutions have pH values less than 7 (3) Basic solutions have pH values greater than 7 e. Most biological substances have pH values between 6 to 8, therefore neutral. (1) homeostasis requires optimum pH ranges ex. Acid Rain (2) Buffers resist change in pH when H+ or OH- are added a. buffers accept H+ ions from the solution when they are in excess and donate H+ when they are depleted. b. buffers usually consist of a weak acid and its corresponding base.