Honors Chemistry Study Guide for Chapter 3: Matter
advertisement

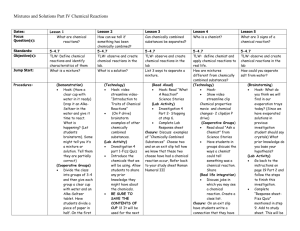
Chemistry 300 Study Guide for: Matter - Properties and Change Matter= anything that has mass and volume (takes up space) Mass= the quantity of matter in an object (also, how much force it takes to accelerate it) Volume= the amount of space occupied by an object I Properties of Matter A. Physical Properties – identifying characteristics that can be determined without changing the composition of the material. Ex. – boiling point, volume, solubility, color. Sample (how much you have). Ex. – length, quantity of heat, mass, volume. B. Chemical Properties – identifying characteristics of a material that can only be determined by an attempt to change the composition of the material; describes the ability to be changed, not the actual process of changing. Ex. – flammability, oxidation, reaction with acids. C. States of Matter – physical form of materials. 1. Solids – state of matter that has both a definite volume and a definite shape. 2. Liquids – state of matter that can flow and has a definite volume but assumes the shape of its container. 3. Gases – state of matter that can flow and has neither a definite volume nor a definite shape. II Changes in Matter A. Physical Changes – alteration to materials in which shape, number of pieces, or state is different but the identifying characteristics remain the same. Ex. - melting, bending, breaking, dissolving. B. Chemical Changes – alteration to materials in which the composition of the material is different; a new material is produced; describes the actual process of change. Ex. - burning, decomposing, explosions, rusting, any reaction which makes a new material. C. Signs of a Chemical Change (What to Look for in Labs): a. Change in color b. Change in odor c. Production of a new substance i. Gas (seen as bubbles) ii. Solid (seen as a “precipitate” or other new, solid substance) d. Energy change that did not result from adding or removing heat/energy manually i. An increase in temperature indicates that heat was released by the reaction to the environment (exothermic) ii. A decrease in temperature indicates that heat was absorbed by the reaction from the environment (endothermic) IV Classifying Matter Pure substance = material that cannot be physically separated into its components. Elements and compounds (molecules and ionic compounds) are pure substances. Mixture = material that can be physically separated into its components A. Mixtures – type of matter that contains two or more materials that are not chemically bonded and are not in a definite ratio (proportion). 1. Heterogeneous Mixtures – Composition is NOT the same throughout. Matter that contains two or more materials not chemically bonded, and has visually distinguishable parts. Ex. – soil, vegetable soup, beach sand. 2. Homogeneous Mixtures (Solutions) – Composition is the same throughout. Matter that contains two or more materials not chemically bonded, and has no visually distinguishable parts. Ex. – soda, air, brass. Separating Mixtures – techniques or processes for isolating one component of a mixture from the others. a. Filtration – separation technique that uses a porous barrier to isolate a solid from a liquid. b. Evaporation – separation technique removes the liquid from a solution as a vapor. B. Elements – homogeneous type of matter that consists of only one kind of atom; cannot be further separated into simple substances by ordinary physical or chemical means. Ex. – iron, neon, carbon. C. Compounds – homogeneous type of matter that consists of two or more elements, chemically bonded, in a definite ratio. Can be chemically separated into elements. They are combined in fixed and regular amounts, with specific formulas. Ex. – water, sodium chloride, carbon dioxide (CO2). 3.