Esiste oggi un forte impulso in campo farmaceutico, agrochimico ed
advertisement

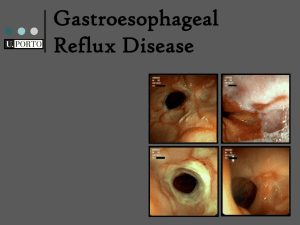
COMPLEXATION STUDIES OF THE PROTON PUMP INHIBITORS WITH NATURAL AND MODIFIED CYCLODEXTRINS L. Marzocchi1, A. Rossi1, J.R. Moyano2, R. Bettini1, A. Gazzaniga3, F. Giordano1 1 Pharmaceutical Department, Faculty of Pharmacy, University of Parma, Italy 2 Department of Pharmacy and Pharmaceutical Technology, Faculty of Pharmacy, University of Seville, Spain 3 Institute for Pharmaceutical Chemistry , University of Milan, Italy The class of drugs commonly referred to as the proton pump inhibitors (PPIs) is used in the systemic treatment of many pathologies related to a hypersecretion of gastric acid. By covalently binding certain critical cystein residues, present in the extracellular luminal domain of the proton pump protein, the PPIs block the conformational reassessments responsible for the protein’s pumping action, and thus inhibit the extrusion of protons into the lumen of the stomach 1,2. However, an initial protonation of the PPI prodrug molecule itself is necessary. The pharmacological potential of these compounds is currently harnessed in gastro-resistant oral formulations which guarantee them a systemic effect and a localised protonation in the secretory canalicula of the gastric mucosa. Whereas localised protonation triggers off biological activity in vivo, the same series of events leads to their decomposition in vitro, making these compounds difficult to manage during drug formulation. The sole desire for greater stability and the welcomed prospect of better bioavailability make PPIs perfect candidates for complexation with cyclodextrins3. These compounds (omeprazole and derivatives) share the common chemical structure of a benzimidazole moiety linked, via a methylensulfinyl bridge, to a pyridine ring and, theoretically, both of these aromatic moieties are able to interact with the cyclodextrin cavity. As part of a more ample research project4-6, the possibility of obtaining inclusion complexes of native and modified cyclodextrins (–cyclodextrin, hydroxypropyl––cyclodextrin and the sodium salt of sulfobutylether –cyclodextrin) with various PPIs , via kneading and freeze-drying has been evaluated. Differential Scanning Calorimetry, Fourier Transform Infrared Spectroscopy, and X-ray Diffractrometry on powder were used to characterise the resulting solid phases. Furthermore, 1H NMR spectroscopy was chosen as the analytical technique to follow the supramolecular interactions residing at the heart of inclusion complex formation in solution; the continuous variations (Job’s Plot) and the phase-solubility7 methods were adopted to determine the stoichiometry of the resulting complexes. References 1. Besancon, M.; Simon, A.; Sachs, G. et al. Sites of Reaction of the Gastric H+,K+-ATPase with Extracytoplasmic Thiol Reagents. J. Biol. Chem., 1997, 22, 438. 2. Sachs, G.; Shin, J.M.; Briving, C. et al. The Pharmacology of the Gastric Acid Pump: the H+,K+-ATPase. Ann. Rev. Pharmacol. Toxicol., 1995, 35, 277. 3. Szejtli, J. In Cyclodextrin Technology. Davies, J.E.D. Ed.; Kluwer Acad. Pub., Dordrecht-Boston-London, 1998. 4. Arias, M.J.; Muñoz, P.; Moyano, J.R.; Ginés, J.M.; Novak, Cs. Thermal Studies of Different Omeprazole/-CD CoGround Systems. J. Thermal Anal., 1998, 51, 973. 5. Arias, M.J.; Moyano, J.R.; Muñoz, P.; Ginés, J.M.; Justo, A.; Giordano, F. Study of Omeprazole/-Cyclodextrin Complexation in the Solid State. Drug Dev. Ind. Pharm., 2000, 26, 253. 6. Marzocchi; L.; Moyano, J.R; Rossi, A. ; Muñoz, P.; Arias, M.J; Giordano, F. Current Status of ATP-ase Proton Pump Inhibitor Complexation with Cyclodextrins. Biolog. J. Armenia, 2001, 176. 7. Higuchi, T; Connors, K.A. Adv. Anal. Chem. Instr. 1965, 4, 117.