Common Ions Table: Chemistry Reference
advertisement

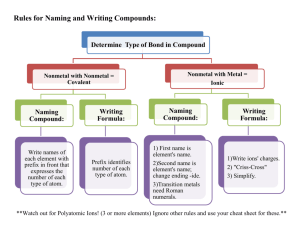
TABLE OF COMMON IONS (You need to memorize the ones in bold type.) POSITIVE IONS (Cations) +1 ammonium (NH4+) cesium (Cs+) copper (I) (Cu+) gold(I) (Au+) hydrogen (H+) lithium (Li+) potassium (K+) rubidium (Rb+) silver (Ag+) sodium (Na+) NEGATIVE IONS (Anions) -1 acetate (C2H3O2- or CH3COO-) bromate (BrO3-) bromide (Br-) chlorate (ClO3-) chloride (Cl-) chlorite (ClO2-) cyanate (CNO-) cyanide (CN-) dihydrogen phosphate (H2PO4-) fluoride (F-) hydrogen carbonate (HCO3-) hydrogen sulfate (HSO4-) hydrogen sulfite (HSO3-) hydroxide (OH-) hypochlorite (ClO-) hypobromite (BrO-) iodate (IO3-) iodide (I-) nitrate (NO3-) nitrite (NO2-) perchlorate (ClO4-) periodate (IO4-) permanganate (MnO4-) thiocynate (SCN-) +2 barium (Ba2+) beryllium (Be2+) cadmium (Cd2+) calcium (Ca2+) chromium (II) (Cr2+) cobalt (II) (Co2+) copper (II) (Cu2+) iron (II) (Fe2+) lead (II) (Pb2+) magnesium (Mg2+) manganese (II) (Mn2+) mercury (I) (Hg22+) mercury (II) (Hg2+) nickel (II) (Ni2+) platinum (II) (Pt2+) strontium (Sr2+) tin (II) (Sn2+) zinc (Zn2+) +3 aluminum (Al3+) chromium (III) (Cr3+) cobalt (III) (Co3+) gallium (Ga3+) gold (III) (Au3+) iron (III) (Fe3+) nickel (III) (Ni3+) +4 lead (IV) (Pb4+) manganese (IV) (Mn4+) platinum (IV) (Pt4+) tin (IV) (Sn4+) -2 carbonate (CO32-) chromate (CrO42-) dichromate (Cr2O72-) hydrogen phosphate (HPO42-) oxalate (C2O42-) oxide (O2-) peroxide (O22-) selenide (Se2-) silicate (SiO32-) sulfate (SO42-) sulfide (S2-) sulfite (SO32-) telluride (Te2-) thiosulfate (S2O32-) -3 arsenate (AsO43-) borate (BO33-) nitride (N3-) phosphate (PO43-) phosphide (P3-) Use the following discussion notes to help you learn the names and formulas of monatomic and polyatomic ions. Monatomic Ions o All monatomic ions in group 1 are (+1) o All monatomic ions in group 2 are (+2) o All nonmetallic monatomic ions in group 15 are (-3) o All nonmetallic monatomic ions in group 16 are (-2) o All monatomic ions in group 17 are (-1) o All metallic monatomic ions in group 13 are (+3) unless the name contains a Roman numeral that indicates the charge. o Most metallic ions in groups 3-16 have multiple charges. The charge on these metals that have multiple monatomic ions will be indicated by using a Roman numeral (in the name only) that is equivalent to the charge. Metals that have only one monatomic ion do not have Roman numerals in their name. Polyatomic ions o All ions to be memorized end in (-ate). Exceptions: hydroxide, peroxide, cyanide. o Three very important ions that you memorized are models for similar polyatomic ions in their corresponding groups. These ions are limited to Groups 15-17 and periods 3, 4 and 5. Note that the charges on these ions are the same as the groups listed above. Group 15 - Phosphate (arsenate, antimonate) , XO43 Group 16 - Sulfate (selenate, tellurate) , XO42 Group 17 - Chlorate (bromate, iodate) , XO31o All ions that end in (-ite) have a charge equal to the corresponding (-ate) ion but contain one less oxygen atom. o All ions that have one less oxygen atom than those ending in (-ite) have the prefix hypoadded to the (-ite) name. Their charge remains the same as the (-ite). Remember that hypomeans lower or under. o All ions that have one more oxygen atom than those ending in (-ate) have the prefix peradded to the (-ate) name. Their charge remains the same as the (-ate). Per- is an abbreviated form of the word hyper- which means more or over. o (*) Polyatomic ions that have -2 or -3 charges may have one hydrogen ion (H+) added to form a new polyatomic ion. The name and the charge on these new ions are a composite of hydrogen ion and the original polyatomic ion. The hydrogen ion, H+, attached to the carbonate ion, CO32-, to become the hydrogen carbonate ion, HCO31-. Note: It is still common for people to call these new polyatomic ions using the prefix bi- instead of the word hydrogen, bicarbonate instead of hydrogen carbonate. o (*) Polyatomic ions that have -3 charge may have two hydrogen ion (H+) added to form a new polyatomic ion. The name and the charge on these new ions are a composite of the two hydrogen ions and the original polyatomic ion. Two hydrogen ions, 2 H+, attached to the phosphate ion, PO43-, to become the dihydrogen phosphate ion, H2PO41-. (*) Note that these new entities are ions and not compounds. A compound has no charge. When anions (negative ions) combine with hydrogen ions to form compounds, the compounds are generally named as acids.