Ionic Properties:
advertisement

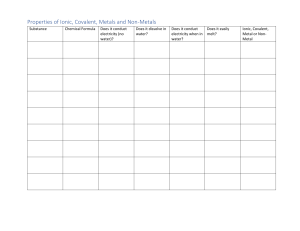
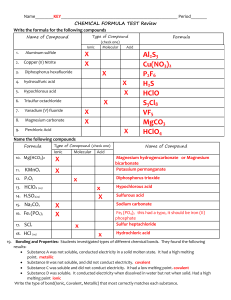
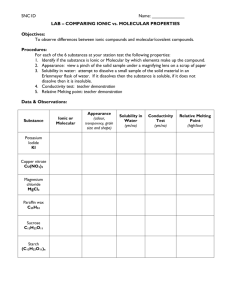
(TP, Ch 4) Ionic Properties: Ionic compounds: are composed of well-organized, tightly bond ions. are crystalline solids at room temperature. are hard, rough and brittle. have high melting points due to the attractions between the positive and negative charges. (above 300°C) dissolve in water and conducts electricity (electrolytes). Electrolytes are compounds that conducts electricity when melted or dissolved in water. KCl, CuSO4 , NaCl, NaHCO3 (salt sub., pools, salt, baking soda) Representative Unit is a Formula Unit Covalent Properties Covalent Molecules: Composed of neutral atoms and held together by strong bonds Weak intermolecular (interparticle) forces causes them to be liquids or gases at room temperature. Do not conduct electricity in a pure state Many do not dissolve in water, some do (sugar); generally less soluble in water Representative Unit is a Molecule Melting point – low (below 300°C) **Table 16.5 p. 465(cp)** (transparency?) 1