ORGANIC CHEMISTRY

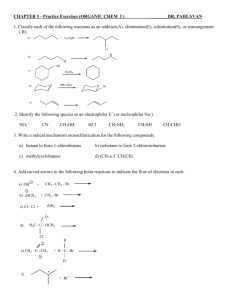
c.
1. a.
ORGANIC CHEMISTRY
WORKSHOP 6
Carboxyl Derivatives II
Provide structures that satisfy the following descriptions:
CH 399, Spring 2003
the structure of the compound, C
8 permanganate to a compound, C
8
H
10
, that is converted upon vigorous oxidation with
H
6
O
4
, which loses water upon heating. b. d. the structure of the compound, C
6
H
10
O
3
, that exhibits strong absorption in the infrared spectrum at 1754 cm –1 and 1818 cm –1 and the following 1 H NMR spectrum:
1.19 (3H, t)) and 2.48 (2H, q). the stereoisomer of 3-hydroxycyclohexanecarboxylic acid that is capable of forming a lactone. the structure of the compound, C
6
H
12
O
2
, that would undergo reaction with excess ethyl magnesium bromide to give the compound shown below.
OH
2.
Propose a plausible mechanism for each of the following reactions. Provide all of the important Lewis structures that contribute to the resonance hybrid for delocalized intermediates.
O a.
C N
HCl
H2O, ²
C OH + NH4
O
O b.
N CH
3
NH
2
NH
2
NH
NH
+ CH
3
NH
2 c.
O
O
C
CH
3
O C
CH
CH
3
3
O = O-18
H
3
O , H O
O
O
C
H
O
+
CH
3
CH
3
C OH + CH
2
CH
3
C
CH
3
CH
3 d.
(Compare the mechanism he re with that of a more typic al ester hydrolysis, and a ccount for any difference s.)
O
1. CH
3
CH
2
Li
O O
2. H
3
O
OH +
HO
OH
c.
f.
a.
3. Esters of mesitoic acid (below) are notoriously difficult to hydrolyze, and require a special procedure. This involves dissolving the ester in concentrated sulfuric acid, then pouring the acid solution into cold water or over crushed ice. What causes the ester to be so resistant to hydrolysis, and what might be the mechanism operating here?
O OCH
C
2
CH
3
O OH
C
H
3
C CH
3
1. c onc. H
2
SO
4
H
3
C CH
3
+ CH
3
CH
2
OH
2. H
2
O
CH
3 ethyl me sitoate
CH
3
4. Give the structure(s) of the major organic product(s) for the following reactions.
O b.
O 1. CH3MgBr
2. NH4Cl, H2O
OH
CH3CHCH2CH3 + PhN C O d.
e.
g.
h.
1.
CH
2
MgCl
CH
2
C N
2. H
3
O
+
O O O O
NHCH
2
CH
2
NH CCH
2
CH
2–
C
–
NHCH
2
CH
2
NH C
–
CH
2
CH
2
C n
O
OH
Cl Cl
(C
7
H
4
O
3
)
OH
CO
2
CH
2
CH
3
1. LiAlH
4
2. H
3
O
O
O
2
N
OCH
3
H
2
, Pt
C
6
H
11
NO
CH
3
O
O
C
O
C OCH dimethyl terephthalate
3
+ HO CH
2
CH
2
OH ethylene glycol ac id
HCl
H2O, ²
Dacron polyester
O ex cess
NH i.
Cl Cl