Rocks & Building Materials
advertisement

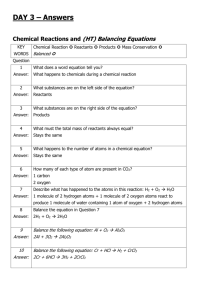
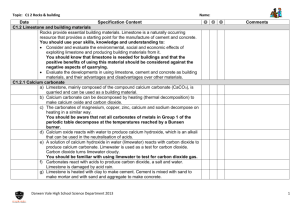
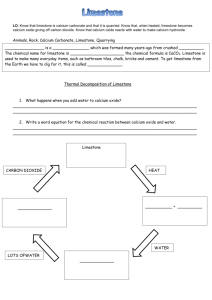
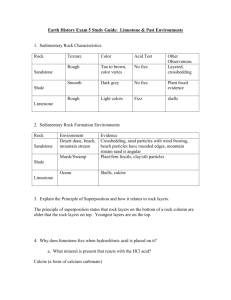
CALCIUM CARBONATE Calcium carbonate (CaCO3) is an important raw material which occurs naturally as the sedimentary rocks, limestone and chalk, and as the metamorphic rock, marble. These rocks are obtained by quarrying. Malham cove (limestone) Dover cliffs (chalk) marble quarry Thermal Decomposition When some substances are heated, they decompose (break down) into simpler substances. This process is called THERMAL DECOMPOSITION. Calcium carbonate undergoes thermal decomposition when heated in a kiln to about 1100 oC, forming calcium oxide (quicklime) and carbon dioxide. Other metal carbonates decompose in a similar way. The lower the metal is in the reactivity series, the lower the temperature at which this decomposition takes place. CaCO3(s) CaO(s) + CO2(g) Metal carbonates such as zinc and copper carbonates therefore decompose easily whilst sodium and potassium carbonates do not. Sometimes we can tell that thermal decomposition has occurred by a colour change (eg green copper carbonate turns black). CuCO3(s) CuO(s) + CO2(g) However if there is no colour change we can test for the production of CO2 gas using limewater. If CO2 is given off the limewater will turn cloudy and decomposition has occurred. Test for CO2 When CO2 reacts with limewater the following reaction occurs which produces a white insoluble precipitate (which makes it cloudy) Ca(OH)2 (aq) + CO2 (g) Calcium hydroxide + carbon dioxide (limewater) TOPIC 10.1.2: ROCKS & BUILDING MATERIALS 1 CaCO3(s) + H2O (l) Calcium carbonate + water Hydration Quicklime reacts with water in a very exothermic reaction, forming calcium hydroxide (slaked lime). CaO(s) + H2O(l) Ca(OH)2(s) The Limestone Cycle H2O LIMESTONE (calcium carbonate) CO2 CO2 QUICKLIME (calcium oxide) SLAKED LIME (calcium hydroxide) H2O Uses of Calcium Carbonate & Calcium Hydroxide Building Material Limestone is used as a building material. A form of limestone, called Portland stone, was used in the construction of St Paul’s Cathedral in London. Limestone is a hard rock, but it erodes badly in polluted urban environments, because it dissolves readily in acids. Marble is used in sculpture and as a building material. St. Paul’s Cathedral TOPIC 10.1.2: ROCKS & BUILDING MATERIALS 2 marble bust Removal of Acidity Powdered limestone is used to neutralise acidity in lakes and in light, sandy soils. Slaked lime (calcium hydroxide) is used to neutralise acidity in heavy soils and in drinking water. Glass Manufacture Glass is made by heating a mixture of: limestone sand sodium carbonate Iron production Limestone is added to the Blast Furnace, in the production of iron, to remove acidic impurities present in iron ore. Cement Manufacture Cement is made by roasting powdered limestone with powdered clay in a rotary kiln. Mortar which is used to build a modern house today is a mixture of cement, sand and water. The hard, stone-like building material, concrete is made by mixing: cement water sand crushed rock The soft mixture undergoes a slow chemical reaction and eventually sets hard. TOPIC 10.1.2: ROCKS & BUILDING MATERIALS 3 NEUTRALISATION GLASS heavy soils drinking water SLAKED LIME lakes light soils add water QUICKLIME heat + sand & sodium carbonate heat LIMESTONE CHALK MARBLE CALCIUM CARBONATE heat + powdered clay BUILDING removal of impurities IRON & STEEL TOPIC 10.1.2: ROCKS & BUILDING MATERIALS 4 CEMENT CONCRETE mix with water sand crushed rock END-OF-TOPIC TEST 1. Complete the following sentence by filling in the gaps. The early atmosphere of the Earth consisted mainly of ........................................., like the present-day atmosphere on the planet ............................................... This atmosphere was formed by ..................................................................................... 2. When plants evolved, the atmosphere on Earth began to change. Describe the changes which took place and explain how they enabled life to establish itself on land. .............................................................................................................................…………. ..............................................................................................................................………… ..............................................................................................................................………... .............................................................................................................................……….... 3. The carbon cycle keeps the amount of carbon dioxide in the atmosphere almost constant. State THREE ways in which carbon dioxide is released into the atmosphere. .................................................................................................................………….. .................................................................................................................………..... .................................................................................................................………..... State THREE ways in which carbon dioxide is removed from the atmosphere. ....................................................................................................................……….. ....................................................................................................................……….. ....................................................................................................................………... 4. Which gas is the main component of the Earth’s present-day atmosphere? How was this gas formed? ..............................................................................................................................………... ..............................................................................................................................………... TOPIC 10.1.2: ROCKS & BUILDING MATERIALS 5 5. Give the names of two naturally occurring forms of calcium carbonate, one a sedimentary rock, the other a metamorphic rock. Sedimentary ........................................................................................................……….... Metamorphic ........................................................................................................………... Complete this sentence: When calcium carbonate is heated strongly, it undergoes a chemical reaction called .......................................................................... and forms a gas called ............................................................ and a solid called ......……........................... This solid is also known as ........................................................................... Describe what you would see when water is added to this solid. ..............................................................................................................................………... 6. How is calcium carbonate turned into cement? ..............................................................................................................................……….... Which other substances are added to cement to make concrete? ..............................................................................................................................………... State THREE uses for calcium carbonate, other than in cement-making. ............................................................................................................................….. .............................................................................................................................…. ..............................................................................................................................… 7. What is the main reason for the rise in the average global temperature? ..............................................................................................................................………... Describe the effect that this rise could have in Greenland and in Holland. Greenland ............................................................................................................……….... .Holland ................................................................................................................………... Describe one way in which we could attempt to reverse this change in climate. ..........................................................................................................................………....... ...............................................................................................................................…………………. TOPIC 10.1.2: ROCKS & BUILDING MATERIALS 6