Approved objectives Completed
advertisement

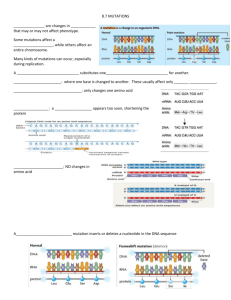
HIGHER EDUCATION COMMISSION NATIONAL RESEARCH PROGRAM OF UNIVERSITIES FINAL REPORT Reporting period from: June, 2003 to: June, 2006 HIGHER EDUCATION COMMISSION NATIONAL RESEARCH PROGRAM OF UNIVERSITIES FINAL REPORT PART: 1 GENERAL INFORMATION TITLE OF PROJECT: Screening for Vision and Hearing Impairment in Rural Sindh and Balochistan REPORTING PERIOD FROM : June, 2003 PRINCIPAL INVESTIGATOR’S NAME: To: June, 2006 Sheikh Riazuddin PRINCIPAL INVESTIGATOR’S INSTITUTE MAIL ADDRESS: Centre of Excellence in Molecular Biology, University of the Punjab, 87- West Canal Bank Road, Thokar Niaz Baig, Lahore-53700, Pakistan. TELEPHONE: E-MAIL: 92-42-5421235 FAX: 92-42-5164155 riaz@lhr.comsats.net.pk ABSTRACT: Sixty large consanguineous families comprising four or more affected siblings were enrolled as part of the studies to Screen for Vision and Hearing Impairment in Rural Sindh and Balochistan. The levels of hearing impairment was determined by audiometric analysis and vision impairment was determined by fundoscopy and electroretinography (ERG). Blood samples were collected from affected as well as non-affected siblings and living parents and grand parents. DNA was isolated and processed for linkage analysis for the presence of known DFNB loci and RP loci. Twenty three families were found linked to DFNB1, B2, B3, B4, B7/11, B9, B12, B21, B23, B29 & B37. Ten families were found linked to PDE6A, TULP1, RPE65, CNGB1/BBS2 & CNGA1. Sequencing of the four PDE6A linked families identified three different mutations while in the fourth family linked to the same region, no disease causing mutation was identified in PDE6A gene. Genome wide scan was carried out on selected six unlinked vision impaired families in search for new Retinitis pigmentosa (RP) loci/genes. A new recessive locus mapped to a 14.21 cM (21.19 Mb) region on chromosome 8q11 was identified in three families. Sequencing of the coding exons of RP1 known to cause dominant RP shows mutations in all 3 families. Another new recessive locus RP32 was mapped on chromosome 1 P13.3 – P21.2 that causes severe autosomal recessive RP. Among the DFNB linked families, three DFNB1 families upon sequencing revealed two mutations in the coding exon of GJB2 gene. Forty nine exons of MYO7A gene were sequenced in the five families linked to DFNB2/USH1B and three mutations were identified. Sequencing of 21 exons of SLC26A4 gene in the five families linked to DFNB4/PDS, identified mutations in exons 3, 6 & 19. Genome wide scan was carried out on selected four unlinked hearing impaired families in search for new deafness loci/genes. A new locus DFNB48 mapped to chromosome 15 q 23–25.1 was identified in two families from Sindh province (Ahmad, et al., 2005). The DFNB48 locus acronym was provided by Human Genome Organization Committee (HUGO): http://www.gene.ucl.ac.uk/hugo. In one family form Balochistan another new locus mapped to chromosome 6p21.1-p22.3 was identified with locus acronym DFNB67. Sequencing of the coding exons of TMHS shows mutation segregating with the hearing phenotype (Shabbir et al., 2006). Normal allele frequencies for the markers in the region of DFNB48 and DFNB67 were calculated by genotyping 90 normal random samples collected from the same province. The above results have contributed to our understanding of the problem of the prevalence/ role of mutations that cause hearing/vision impairment in different ethnicities in Sindh and Balochistan. PART: 2 BACKGROUND / METHODOLOGY BACKGROUND INFORAMTION: Retinitis pigmentosa (RP) is a group of inherited diseases that affect the retina. The disease is characterized by a gradual break down and degeneration of photoreceptor cells (Rods and Cones) that results in a progressive loss of vision. The rate of progression and degree of visual loss are variable. The symptoms of RP include night blindness, loss of peripheral vision and ability to discriminate colors. RP is classified as non syndromic and syndromic. Non syndromic RP may be familial (Multiplex) or isolated (Simplex). Mode of inheritance is Autosomal dominant, Autosomal recessive, X-linked, and Mitochondrial. To date 47 non syndromic loci have been localized of which 37 genes are known. The spectrum of hereditary deafness is broad and ranges from simple deafness without other clinically recognizable abnormalities (non-syndromic) to genetically determined syndrome of a more pleiotropic nature. Syndromic form with anomalies of the eye (Usher Syndrome) is the most common syndrome associated with hearing impairment (Bergstorm, et al., 1971). A number of mutated genes have been identified that are responsible for syndromic deafness, associated with vision impairment and it is believed that efforts for search for new genes / loci is not at all completely exhausted. Localization of genes responsible for recessive hearing / vision loss is often difficult because of the extreme heterogeneity of these genetic disorders and small family sizes in many countries (petit, 1996). Besides locus heterogeneity, allelic heterogeneity is common. The allelic heterogeneity is sometimes associated with clinical heterogeneity, with non-syndromic hearing / vision impairment being caused by mutations within the same gene. Furthermore, different mutations within the same gene result in a variety of phenotypes e.g mutation of MYO 7A can cause Usher syndrome USH1B, dominant DFNA11and recessively inherited non-syndromic deafness DFNB2 (Liu, et al 1997, Weil, et al., 1997). In a background of the above, it was proposed to expand the scope to extend the ongoing studies on hearing / vision impairment to scan different ethnicities in interior Sindh and Balochistan. A single inbred family may have several deaf / vision impaired individuals, which will be helpful in linkage analysis as well as cloning of the newly discovered genes. The over all objective of the proposed studies is to obtain information on the prevalence of mutations in genes as well as new loci involved in hearing / vision impairment. WORK PROGRESS/ RESULTS Sixty large consanguineous families (35 hearing and 25 vision impaired) having four or more affected individuals were enrolled in this programme. Detailed medical history with any observable clinical symptom was recorded. Audiograms of selected deaf individuals, their parents and siblings were conducted. An ophthalmologist examined those individuals with a history of vision impairment for fundoscopy and field analysis. In certain selected cases, the affected individuals were transported to CEMB to confirm the diagnosis of retinitis pigmentosa by Electro-retinography (ERG). Blood samples were collected and genomic DNA was extracted by standard methods. Genomic DNA was analyzed by genotyping for known DFNB loci. Twenty three hearing impaired pedigrees were identified as linked to reported loci (Table-1). TABLE – 1 : DEAFNESS LOCI DFNB 1 DFNB 2 DFNB 3 DFNB 4 DFNB 7/11 DFNB 9 DFNB 12 DFNB 21 DFNB 23 DFNB 29 DFNB 37 Total NO. OF LINKED PEDIGREES 3 5 2 5 1 1 1 1 1 1 2 23 Sequencing for GJB2 (DFNB1) It has been reported that hearing impairment that cosegregates with DFNB1, is caused by mutations in GJB2 gene (Guilford et al. 1994). GJB2 gene is located on chromosome13 at position 13q11and has two exons. Exon 1 encodes the 5’ untranslated region and exon 2 contains the entire open reading frame (680 bp) encoding 208 amino acid long protein product called connexin26 (Cx26). Coding exon 2 was sequenced in the three linked families and the results are shown below. Exon 2 2 Mutation W77X W24X Sequencing for MYO7A (DFNB2/USH1B) DFNB2/Usher 1B is located on chromosome 11 at 11q13.5 and includes 49 exons (Weil, D. et al.1997). . All the 49 exons in five linked families were sequenced. The following three mutations were identified: Exon Mutation 7 G214R 21 A826T 28 E1170K Sequencing for SLC26A4 (DFNB4/PDS) SLC26A4 gene lies on chromosome 7 at 7q31 (Baldwin et al. 1995). SLC26A4 has 21 exons. All exons in the five linked families were sequenced and three mutations were identified. Exon 3 6 19 Mutatiom S90L V239D K715N Genome wide scan was carried out on selected four unlinked hearing impaired families in search for new deafness loci/genes. A new locus DFNB48 mapped to chromosome 15 q 23–25.1 was identified in two families from Sindh province (Ahmad, et al., 2005). The DFNB48 locus acronym was provided by Human Genome Organization Committee (HUGO): http://www.gene.ucl.ac.uk/hugo. In one family form Balochistan another new locus mapped to chromosome 6p21.1-p22.3 was identified with locus acronym DFNB67. Sequencing of the coding exons of TMHS shows mutation segregating with the hearing phenotype (Shabbir et al., 2006). Normal allele frequencies for the markers in the region of DFNB48 and DFNB67 were calculated by genotyping 90 normal random samples collected from the same province. When genomic DNA of vision impaired families were analyzed by genotyping for known RP loci, the ten families were found as linked to reported loci (Table-2) TABLE – 2 : RP LOCI PDE 6A TULP1 RPE65 CNGB1/BBS2 CNGA1 Total NO. OF PEDIGREES 4 2 2 1 1 10 Genome wide scan was performed on selected six pedigrees unlinked to the reported RP loci. In one family linkage was detected on chromosome 1p13.3-p21.2 defining a novel locus RP32. This new locus resides on chromosome 1 between D1S2896 and D1S457 & causes severe autosomal recessive RP. Another linkage was detected on chromosome 8q11 in one family defining a novel locus of Retinitis pigmentosa. In order to confirm this linkage and fine map the genetic interval, additional families present in CEMB repository were screened and two more families were linked to the same region. RP in all 3 families mapped to a 14.21 cM (21.19 Mb) region on chromosome 8q11 flanked by D8S532 and D8S260. This region harbors RP1, which is known to cause autosomal dominant retinitis pigmentosa. Sequencing of the coding exons of RP1 shows mutations in all 3 families: two single base deletions, c.7470delA and c.8168delA resulting in a frame shift and a 4bp insertion c4377-4378insTGAA all causing premature termination of the protein. Rp in four families maps to a 13.85 cM (14.87 Mb) region on chromosome 5q31-33 flanked by D5S2090 and D5S422. This region harbors the PDE6A gene, which is known to cause arRP. Sequencing of PDE6A shows a single base pair change c.889→ T, a single base pair insertion c.2218→ 2219 ins T and a single base pair substitution in the splice accepter site IVS 10-2 A→G in three families. In the fourth family no disease causing mutation was identified in the PDE6A gene. OBJECTIVES / ACHIEVEMENTS Approved objectives Completed Search for families in rural Sindh & Balochistan with 3 or more hearing and vision impaired individuals. 100% Collection of blood samples from the affected and close relatives in the pedigree and DNA Extraction from affected and normal individuals. 100% Clinical tests, Audiograms, Fundoscopy, Electroretinography (ERG) to identify the type of deafness and vision impairment in the family. 100% Genotyping for screening the reported loci and search for new loci. 100% Mutation screening. 100% METHODOLOGY Blood and Buccal Swab Collection Blood samples (10cc) were collected from parents, affected children and normal siblings of each nuclear family along with all the available elder members of the Pedigree. EDTA was used as an anticoagulant. Blood was kept at 4OC before processing for DNA extraction. In case of elderly or small children buccal swabs (Epicentre Technologies WI, Medical Package Corporation, CA, USA) were used to collect buccal epithelial cells. DNA Extraction Blood samples were processed to extract DNA by a non-organic method (Grimberg et al., 1989). DNA concentrations were determined by spectrophotometer. DNA from buccal swabs was extracted according to the instructions of manufactures Epicentre Technologies WI, Medical Package Corporation, CA, USA. Linkage analysis for known DFNB & RP loci Efforts were made to ascertain the families, which fall into the already known linkage region. For this purpose, reported DNFB & RP markers were used. The microsatellite markers were amplified by polymerase chain reaction, using genomic DNA as a template. These markers are listed in genome data base (GDB) and only fluorescent markers were used. The PCR product of the markers were pooled in such a way that the marker size and their dye do not overlap. The PCR products were analyzed on the ABI 3100 genetic analyzer and alleles were assigned using Genescan and Genotype Software. Scans showing various allele patterns for the microsatellitc markers, were labeled and placed on the corresponding pedigrees to find out whether the marker cosegregates with the disease gene. In this way the families that link to the known loci were ascertained and those that do not link to any of the known locus were separated. Mutational Analysis BigDyeTM Terminator Cycle Sequencing Ready Reaction Kit (Perkin Elmer/Applied Biosystems) was used to sequence the PCR products (RP1). The thermal cycling was done in GeneAmp PCR system 9700 & 2700 (Perkin Elmer). 10μl deionized formamide (Perkin Elmer) was used to suspend the pellet and loaded on ABI 3100 genetic analyzer/sequencer according to the manufacturer’s instructions. Any change in the DNA sequence was confirmed by sequencing both sense and antisense strands. Once a mutation is confirmed, samples from all available affected individuals and their normal siblings were also sequenced to confirm that the change in the DNA sequence is segregating with affected phenotype. Genome wide scan Genome-wide search was undertaken using 358 fluorescently labeled microsatellite markers at an average spacing of 10 cM (ABI, Prism 1, Linkage mapping set 2.5, Applied Biosystems). 2 µl of the PCR product along with 11.8 µl of formamide and 0.2 µl LIZ size standard (Applied Biosystems) was pooled for genotyping. Amplified products were resolved by capillary electrophoresis using an ABI Prism 3100 Genetic Analyzer. The alleles were assigned using Genescan and Genotyper software (Applied Biosystems). Lod score calculation For linkage analysis, the Marshfield genetic map was used for marker order and map distances. Lod scores were calculated by using MLINK of the FASTLINK computer package. Disease was coded as fully penetrant and disease allele frequency was set at 0.001. Meiotic recombination frequencies were assumed to be equal for males and females. PUBLICATIONS 1. Ahmad J., Khan S N., Khan S. Y., Ramzan K., Riazuddin S., Ahmed Z M., Wilcox E R., Friedman T B., Riazuddin S. 2005. DFNB48, a new Nonsyndromic recessive deafness locus maps to chromosome 15q23-q25.1. Hum Genet. 116:407-412. 2. S.Amer Riazuddin, Fareeha Zulfiqar, Qingjiong Zhang, Yuri V.Sergeev Zaheeruddin.A.Qazi, Tayyab Hussnain, Raphael Caruso, Sheikh Riazuddin, Paul. A. Sieving, J. Fielding Hejtmancik. 2005. Autosomal Recessive Retinitis pigmentosa is associated with mutations in RP1 in three consanguineous Pakistani families. Investigative Ophthalmology & Visual Science (IOVS). 46(7): 2264-2270. 3. Qingjiong Zhang, Fareeha Zulfiqar, Xueshan Xiao, S. Amer Riazuddin, Muhammad Farooq Sabar, Raphael Caruso,Paul A. Sieving, Sheikh Riazuddin, J. Fielding Hejtmancik 2005. Severe recessive retinitis pigmentosa maps to chromosome 1p13.3p21.2 between D1S2896 and D1S457 but outside ABCA4. Hum Genetics. 28:1-10. 4. Shabbir M I, Ahmed Z M, Khan S Y, Riazuddin S, Waryah A M,Khan S N, Camps R D, Ghosh M, Kabra M, Belyantseva I A, Friedman T B, Riazuddin S. 2006. Mutations of human TMHS cause recessively inherited nonsyndromic hearing loss. J Med Genet. 43(8): 634-40. 5. S.Amer Riazuddin, Fareeha Zulfiqar, Qingjiong Zhang, Wenliang Yao, Shouling Li, Xiaodong Jiao, Amber Shahzadi, Muhammad Amer, Muhammad Iqbal,Tayyab Husnain, Paul Sieving, Sheikh Riazuddin, J. Fieldingg Hejmancik. 2006 Mutations in the gene encoding the α–subunit of rod & phosphodiesterase in Consanguineous Pakistani families. Molecular vision 12:1283-1291. BACKGROUND / METHODOLOGY continued REFERENCES Ahmad J., Khan S N., Khan S. Y., Ramzan K., Riazuddin S., Ahmed Z M., Wilcox E R., Friedman T B., Riazuddin S. 2005. DFNB48, a new Nonsyndromic recessive deafness locus maps to chromosome 15q23-q25.1. Hum Genet. 116:407-412. Bergstorm, L et al., (1971): A high risk registry to find congenital deaness. Otolaryngol Clin North Am; 4: 369 -99. Grimberg, J. et al (1989). A simple and efficient non organic procedure for the isoaltion of genomic DNA from blood. Nucleic Acid. Res. 17: 8390-8391. Liu, et al. (1997). Muations in the Myosin VIIa gene cause non-syndromic recessive deafness, Nat Genet:16:188-190. Petit, (1996). Genes responsible for hereditary deafness - symphony of a thousand Nat Genet 14: 385-9. Shabbir M I, Ahmed Z M, Khan S Y, Riazuddin S, Waryah A M, Khan S N, Camps R D, Ghosh M, Kabra M, Belyantseva I A, Friedman T B, Riazuddin S. 2006. Mutations of human TMHS cause recessively inherited nonsyndromic hearing loss. J. Med. Genet. Electronically published February 2006. Weil, D. et al. (1997). The autosomal recessive isolated deafness DFNB2 and the Usher 1B are allelic defects of the myo VIIa gene. Nat Genet 16: 191-3. SUMMARY Sixty large consanguineous families comprising four or more affected siblings were enrolled as part of the studies to Screen for Vision and Hearing Impairment in Rural Sindh and Balochistan. The level of hearing impairment was measured by pure tone audiometry and vision impairment was determined by fundoscopy and electroretinography (ERG). Blood samples were collected from affected as well as un-affected siblings and living parents and grand parents. DNA was isolated and processed for linkage analysis for the presence of known loci. Twenty three families were found linked to known deafness loci and ten families were found linked to blindness loci. DNA sequencing studies identified different pathological mutations in Sindhi or Balochi people (Riazuddin et al., 2006). Genome wide scan was carried out on selected six unlinked vision impaired families in search for new Retinitis pigmentosa (RP) loci/genes. A new recessive locus mapped to a 14.21 cM (21.19 Mb) region on chromosome 8q11 was identified in three families. Sequencing of the coding exons of RP1 known to cause dominant RP shows mutations in all 3 families (Riazuddin et al., 2005). Another new recessive locus RP32 was mapped on chromosome 1 P13.3 – P21.2 that causes severe autosomal recessive vision impairment. Genome wide scan was carried out on selected four unlinked hearing impaired families in search for new deafness loci/genes. A new locus DFNB48 mapped to chromosome 15 q 23–25.1 was identified in two families from Sindh province (Ahmad, et al., 2005). The DFNB48 locus acronym was provided by Human Genome Organization Committee (HUGO): http://www.gene.ucl.ac.uk/hugo. In one family form Balochistan another new locus mapped to chromosome 6p21.1-p22.3 was identified with locus acronym DFNB67. The results of the above studies have a strong bearing on our understanding of the processes of hearing and vision and the prevalence/ role of mutations that cause hearing/vision impairment in different ethnicities in Sindh and Balochistan. Information on carrier status was provided to the affected families on request for genetic counseling. This information will inevitably help to reduce the incidence of deafness and blindness resulting from high consanguinity in these families. The results of these studies have been published in five peer reviewed international journals.