Strong and Weak Acids/Bases
advertisement

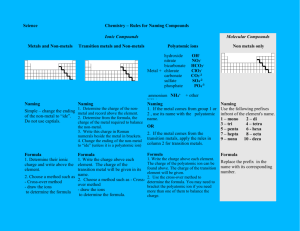
Intermediate 2 Chemistry – Learning Objectives UNIT 3 – ACIDS, BASES AND METALS (a) Acids and Bases The pH scale State that the pH scale is a continuous range from below 0 to above 14. State that acids have a pH below 7, alkalis have a pH above 7 and neutral solutions (such as water) have a pH equal to 7. State that non metals burn to produce non metal oxides. State that non metal oxides which dissolve in water produce acidic solution with the exception of hydrogen oxide which is neutral. Give examples of causes of acid rain including the production of sulphur dioxide from burning contaminated fossil fuel and the production of nitrogen dioxide from the spark in a petrol engine. State that metals react with oxygen to produce metal oxides. State that metal oxides which dissolve in water produce alkaline solutions. State that ammonia is a bitter strong smelling gas which is very soluble in water and forms an alkaline solution. Give examples and uses of everyday acids and alkalis. Give names and formula for laboratory acids and alkalis. Show that acids and electricity conduct electricity and conclude that they must contain free ions in solution. State that hydrogen gas is produced at the negative electrode when acids are electrolysed and conclude that acids must contain hydrogen ions free in solution. State that acids have a greater number of hydrogen ions (H+(aq)) than hydroxide ions (OH-(aq)). State that alkalis have a greater number of hydroxide ions (OH-(aq)) than hydrogen ions (H+(aq)). State that in neutral solutions the number of hydrogen ions is equal to the number of hydroxide ions. H+(aq) = OH-(aq) Explain that when an acid/alkali is diluted, the concentration of H+(aq)/OH-(aq) decreases and the pH moves towards 7. State that water exists in equilibrium between water molecules and hydrogen ions and hydroxide ions. H2O(l) H+(aq) + OH-(aq) State that in equilibrium the concentration of reactants and products is constant but is rarely equal. State that water remains mainly as molecules and therefore is not a good conductor of electricity. Concentration State that a solution is made up by dissolving solute in solvent. State that a concentrated solution has little solvent. State that a dilute solution has much solvent. State that concentration is measured by the number of moles of solute per litre of solvent and therefore has units moles per litre (mol l-1) Calculate number of moles/mass of solute or concentration of solution or volume of solution from the other two variables. Strong and Weak Acids/Bases Explain that strong acids are completely dissociated into ions. (b) HX(aq) H+(aq) + X-(aq) Give hydrochloric, sulphuric and nitric as examples of strong acids. Explain that weak acids are only partially dissociated into ions and mostly remain as molecules. HX(aq) H+(aq) + X-(aq) Give ethanoic and other carboxylic acids as examples of weak acids. Explain strong and weak bases in the same way as acids. Give metal hydroxides as examples of strong bases. Give ammonia solution as an example of a weak base. State that a solution of a strong acid will have a lower pH, higher conductivity and faster reaction rate than an equimolar (same concentration) solution of weak acid. State that a solution of a strong base will have a higher pH, higher conductivity and faster reaction rate than an equimolar (same concentration) solution of weak base. Salt preparation Reactions of acids State that neutralisation of an acid brings its pH towards 7. State that an everyday example of neutralisation is the use of lime to neutralise acidic soil/lakes caused by acid rain. State that substances which neutralise acids are called bases. State that bases which dissolve in water form alkali solutions. State the products of the following neutralisations and write balanced chemical equations for them, including state symbols and identifying spectator ions. I. METAL METAL + ACID SALT + HYDROGEN The hydrogen ions from the acid form molecules of hydrogen gas. The hydrogen gas is identified because it burns with a pop. Acid rain damages metal structures such as iron bridges due to the above reaction. II. METAL OXIDE METAL OXIDE + ACID SALT + WATER The hydrogen ions from the acid and the oxide ions from the metal oxide form molecules of water. III. METAL HYDROXIDE (ALKALI) METAL HYDROXIDE + ACID SALT + WATER The hydrogen ions from the acid and the hydroxide ions from the metal hydroxide form molecules of water. IV. METAL CARBONATE METAL CARBONATE + ACID SALT + WATER + CARBON DIOXIDE GAS The hydrogen ions from the acid and the carbonate ions from the metal carbonate form molecules of water and molecules of carbon dioxide. Acid rain causes damage to marble/limestone buildings/statues because of the above reaction. Acid indigestion can be treated using carbonate indigestion tablets because of the above reaction. Volumetric titrations Calculate unknown volumes or concentrations by titrations with acids or alkalis of known concentration. nH+ x v c = nOH- x v x c Naming Salts State that salts can be formed by the neutralisation of an acid using a base. State that the metal ion from the base will replace the hydrogen ion from the acid to form the salt. State that hydrochloric acid forms chloride salts, sulphuric acid forms sulphate salts and nitric acid forms nitrate salts. State that soluble ammonium salts and nitrate salts are useful as fertilisers because they contain nitrogen. State that when preparing a salt by neutralisation it is easier to use an insoluble base (metal carbonate or metal oxide) so that the end point can be clearly identified. Precipitation State that precipitation is the reaction of two solutions to form an insoluble product called a precipitate. State that insoluble salts can be prepared by precipitation reactions. Ionic equations Write balanced ionic equations for salt formation by precipitation or neutralisation, including state symbols and identifying spectator ions. Rewrite balanced ionic equations for salt formation omitting spectator ions. (c) Metals The electrochemical series State that electricity can be produced by connecting two different metals with an electrolyte to form a simple cell. State that the voltage of the cell varies depending on the pair of metals used and that this is the basis of the electrochemical series. Identify that the flow of electrons will be through the external circuit (wires) from the metal highest in the series to the metal lower in the series. Identify that ion will flow through the internal circuit (salt bridge/solution) to complete the circuit. State that electricity can also be produced when one of the half cells involves a non metal. State that metals atoms high in the electrochemical series will displace metals lower in the electrochemical series when added to a solution of their ions. State that reacting metals with acids will displace hydrogen gas and that this reaction allows the position of hydrogen in the electrochemical series to be established. Redox Reactions State that oxidation is the loss of electrons. Recognise that the reaction of a metal element to form an ionic compound is an example of an oxidation reaction. State that reduction is the gain of electrons. Recognise that metal ions in a compound reacting to form a metal element is an example of a reduction reaction. Write ion electron half equations for oxidation and reduction reactions. Combine oxidation and reduction half equations to write an overall redox equation for a reaction. Identify the products of electrolysis from the oxidation reaction at the positive electrode and the reduction reaction at the negative electrode. Reactions of metals Describe how metals react with oxygen, water and dilute acid. Give an indication of reactivity of the metal given its reactions with oxygen, water and dilute acid. Metal Ores State that native metals are those found uncombined as elements in the earth’s crust. State that ores are naturally occurring compounds of metals found in the earths crust. Explain that less reactive metals are not easily oxidised and are therefore likely to be found as native metals – give examples such as gold and silver. Explain that more reactive metals are easily oxidised and are therefore likely to be found as metal ores. Explain that the extraction of a metal from its ore is an example of a reduction reaction. Explain that less reactive (easily reduced) metals can be extracted from their ore by heat alone – give copper as an example. Explain that some more reactive (less easily reduced) metals must be extracted by heating with carbon / carbon monoxide / hydrogen and that this process is called smelting – give iron as an example. State that iron is extracted from iron ore by large scale smelting in the blast furnace. State that the two main reactions taking place in the blast furnace are the production of carbon monoxide and the reduction of iron oxide. Explain that the most reactive (easily oxidised) metals are extracted from their ores by electrolysis of the molten ore – give aluminium as an example. Corrosion State that corrosion is the chemical reaction of a metal which involves oxidation on its surface. State that during corrosion, the surface of the metal is changed from an element to a compound, metal atoms are changed to metal ions. State that different metals corrode at different rates. State that the corrosion of Iron is referred to as rusting and that the iron compound formed is referred to as rust. State that water and oxygen are required for rusting to take place. Explain that water and oxygen gain the electrons from the oxidation of the metal and form hydroxide ions. State that ferroxyl indicator can be used to detect rust. State that ferroxyl indicator changes from yellow to blue in the presence of iron (II) ions. State that ferroxyl indicator changes from yellow to pink in the presence of hydroxide ions. State that Iron (II) can further oxidise to form Iron (III) ions. State that acid rain and salting the roads in winter cause the rate of rusting to increase. Describe and explain the following corrosion prevention methods; PHYSICAL PROTECTION Paint/oil/grease/plastic provides a physical barrier between the metal and oxygen/water thereby preventing rusting. Electroplating involves coating one metal with another to provide a physical barrier to corrosion however if the barrier is scratched the metal is exposed to corrosion. If the metal is plated with a less easily oxidised metal then a scratch on the surface will speed up the rate of corrosion of the more easily oxidised metal. CHEMICAL (SACRIFICIAL) PROTECTION Coating a metal with another metal higher in the table offers chemical protection from corrosion. The higher metal will oxidise and sacrifice its electrons to the lower metal thus preventing its oxidation and protecting it from corrosion. Chemical protection will be offered even if the coating is scratched. Galvinising is an example of chemical protection of iron by coating with zinc. ELECTRICAL PROTECTION Metal is protected from corrosion by attaching the metal directly to the negative terminal of a power supply thus giving the metal a supply of electrons and preventing its oxidation.