249115_1_Lab_density_15
advertisement

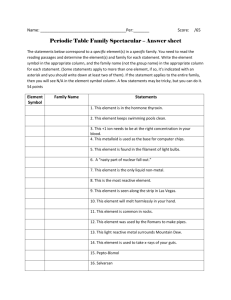
Lab: Density of a metal sample Purpose: Using your measuring and analytical skills you will find the density of 2 metal samples that are actually elements. Different groups will use different amounts of the metals to determine if the density depends on sample size. Finally the calculated density will be used to identify the metals. Hypothesis: Does density depend on the size of the sample. Materials: Balance, 50-mL graduated cylinder, metal samples. Procedure: 1. Obtain metals from your teacher. Mass the sample as directed and record in data table. 2. Find the volume of the object using water displacement: a. Add 20.0 mL of water to graduated cylinder. Check the meniscus carefully. Record the volume in data table as Initial Volume. b. Carefully slide the metal into the grad. cylinder as it rests against the gas nozzle. Record volume in data table as Final Volume, making sure to estimate to the “tenths” place. Have your partner verify the reading. c. Dump the water out of grad cylinder, remove the metal. d. Place metal in designated area. 3. Repeat with second metal sample. Data: Measurements Physical Description Mass of sample (g) Initial Volume (mL) Final Volume (mL) Final Volume – Intial Volume (Objects volume) (mL) Sample 1 Sample 2 Calculations: (Note: Be sure to watch your Sig Figs carefully. Below is an example of how your calculations should look.) (sample calculation) sample 1 D = mass = volume 10.234 g = 5.18 grams/mL 2.00mL sample 2 D = mass = volume 10.234 g = 5.07 g/mL 2.02 mL