6009KB
advertisement

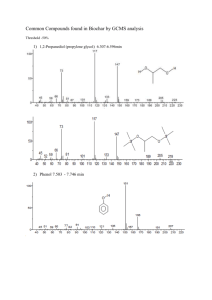
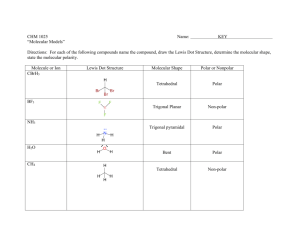
NCEA Level 3 Science (90730) 2011 — page 1 of 4 Assessment Schedule – 2011 Science: Describe selected organic compounds and their uses (90730) Evidence Statement Q ONE Evidence Names of reactants are: Methanol (an alcohol) and octanoic acid (a carboxylic acid). Name of ester formed is methyloctanoate. Structure: Equation (Alcohol + carboxylic acid → ester + water) Methanol + octanoic acid → methyl octanoate + water. Special conditions: Heated under reflux conditions as reactants AND ester are volatile and reflux allows heating for long enough for a good yield of ester to be formed. David Blaker & Sandy Talbett, NCEA Level 3 Science Study Guide (Auckland: ESA, 2005), p 105. Concentrated sulfuric acid used as a catalyst to speed up the reaction and also acts as a dehydrating agent removing the water produced in the reaction. This shifts the equilibrium to the right (favours the forward reaction) resulting in greater yield (amount) of ester produced. Achievement TWO of: • Names reactants. • Names ester. • General word equation. • Draws ester. • Names a condition. Eg: The reactants are methanol and octanoic acid. The equation for an ester is: alcohol + carboxylic acid ester + water Achievement with Merit Draws ester, specific word equation AND Identifies either special condition. Eg: The reactants are methanol and octanoic acid and the ester that is produced is methyl octanoate.. The ester is: The equation for this ester is: methanol + octanoic acid methyl octanoate + water To produce more ester, you can add concentrated sulfuric acid. Achievement with Excellence ALL of: • Draws ester (condensed or structural but must show ester link). • Specific word equation. • Identifies either special condition AND Justifies their use. Eg: The reactants are methanol and octanoic acid and the ester that is produced is methyl octanoate. The equation for this ester is: methanol + octanoic acid methyl octanoate + water To produce more ester, you can add concentrated sulfuric acid. The concentrated sulfuric acid works as a dehydrating agent and removes the water that is produced in the reaction. This then drives the reaction to the products side and therefore increases the yield of ester. NCEA Level 3 Science (90730) 2011 — page 2 of 4 TWO Functional group of double bond circled. Triglyceride structure showing glycerol backbone and 3 ester linkages to long chain fatty acids. Unsaturated alkenes are non-polar and dissolve the non-polar grease and the hydrocarbon chain of the fat molecules. Orange oil contains limonene 90% an unsaturated hydrocarbon. This acts as a solvent for the fat and grease allowing it to be wiped away whereas water, which is polar, does not dissolve the non-polar grease. Identifies functional group. OR Structure of fat molecule described/drawn but must specify ester links and fatty acid (long chain). AND Description of interaction of fats and alkenes. OR Description of effectiveness of limonene on grease. OR Description of effectiveness of water on grease. Eg: Double bond in the limonene structure circled. The limonene cleaning product will mix with the fats and remove them. (polarity not required, accept wrong polarity if consistent, eg limonene is polar so will dissolve polar fat) Identifies functional group. AND Structure of fat molecule described/drawn but must specify ester links and fatty acid (long chain). AND Either: Explanation of interaction of fats and alkenes in terms of polarity. OR Explanation of effectiveness of limonene on grease in terms of polarity. OR Explanation of effectiveness of water on grease in terms of polarity. Eg: Both double bond in the limonene structure circled. The limonene cleaning product will act as a solvent and mix with the fats to remove them. This is because the limonene is non-polar and the fats have a non-polar tail. Like dissolves like. All of: Identifies functional group. Structure of fat molecule described/drawn but must specify ester links and fatty acid (long chain). Explanation of interaction of fats and alkenes in terms of polarity. AND Comparison of effectiveness of limonene on grease compared to effectiveness of water on grease related to polarity of molecules involved. Eg: Both double bond in the limonene structure circled. The limonene cleaning product will act as a solvent and mix with the fats to remove them. The fats have a non-polar tail in their structure, so will only dissolve in a non-polar solvent (like dissolves like). The limonene is nonpolar whereas water is polar. This means that water on its own will not clean the fats as easily as a water limonene mix. NCEA Level 3 Science (90730) 2011 — page 3 of 4 THREE Structure of detergent is long non-polar, hydrophobic (water hating) hydrocarbon chain with polar, hydrophilic (water loving) head. David Blaker & Sandy Talbett, NCEA Level 3 Science Study Guide (Auckland: ESA, 2005), p 138. Action on crude oil, micelle formation. Structure of detergent (labelled diagram or description). OR Action of detergent described. Eg: The detergent has a long nonpolar tail and polar head: Structure of detergent. AND Action of detergent described. Must say micelle and describe the polarity of oil and tail OR water and head. Eg: The detergent has a long non-polar tail and polar head: The detergent mixes with the oil slick and breaks it down into micelles. A micelle has the non-polar tail of the detergent in the non-polar oil, and the polar head sticking out. David Blaker & Sandy Talbett, NCEA Level 3 Science Study Guide (Auckland: ESA, 2005), p 142. After treatment with detergent and solvents the large oil mass is broken down into smaller oil filled micelles which disperse due to wind and wave action (and ultimately sink to the bottom of the ocean where they are digested by microorganisms). NOTE: evidence for Achieve can be taken from Question 2 if the structure of detergent is drawn with reference to limonene. FULL answer. Structure of detergent. AND Action of detergent described. AND Fate of oil given. Eg: The detergent has a long non-polar tail and polar head: The detergent mixes with the oil slick and breaks it down into micelles. A micelle has the non-polar tail of the detergent in the non-polar oil, and the polar head sticking out into the polar water. The oil is surrounded by detergent molecules so the oil slick gets broken up and dispersed through the sea. NCEA Level 3 Science (90730) 2011 — page 4 of 4 FOUR Polymers are made up from monomers. Polyethene is made from monomers of ethene in an addition/polymerisation reaction. David Blaker & Sandy Talbett, NCEA Level 3 Science Study Guide (Auckland: ESA, 2005), p 87. Low density polyethene (LDPE) has branches on its polymer chains that prevent the chains fitting closely, giving a more open structure and a low boiling point because the bonding between chains is not as strong so requires less energy to break these bonds. Flexibility is due to the open structure. High density polyethene (HDPE) has no branched chains in the polymer chains so more intermolecular (therefore stronger) bonding between chains that require more heat energy to break, resulting in higher melting point and less flexibility, more rigid structure. THREE of: • Monomer named. • Reaction named. • Equation given. • Identifies presence of branches in LDPE. Eg: This reaction is called polymerisation and the monomer is ethene. All of: • Monomer and reaction named. • Equation given. • Compares presence of branches in LDPE to lack of branches in HDPE. Eg: This reaction is called polymerisation and the monomer is ethene. The LDPE has branches, whereas the HDPE has no branches, which results in the differing properties. Monomer and reaction named. Equation given AND Compares presence of branches in LDPE to lack of branches in HDPE linked to properties of each. Eg: This reaction is called polymerisation and the monomer is ethene. The LDPE has branches, whereas the HDPE has no branches, which results in the differing properties. The branches in the LDPE mean the polymers don’t pack together as easily, and therefore would have less bonding between chains so less energy is required to break these bonds, resulting in a lower boiling point and an increase in flexibility. The absence of branches in the HDPE mean the polymers pack together more easily, and therefore would result in a higher boiling point and a decrease in flexibility. This is because there would be stronger bonds between the strands that would require more heat energy to break these bonds, therefore a higher boiling point. Judgement Statement Achievement Achievement with Merit Achievement with Excellence 2A 2M 2E