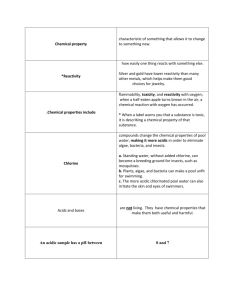
Acids, Bases and Solutions Test
advertisement

Acids, Bases and Solutions Test ____ ____ ____ ____ ____ ____ ____ ____ ____ ____ ____ ____ ____ ____ ____ ____ ____ ____ ____ ____ ____ ____ ____ ____ 1. 2. 3. 4. 5. 6. 7. 8. 9. 10. 11. 12. 13. 14. 15. 16. 17. 18. 19. 20. 21. 22. 23. 24. ____ ____ ____ ____ ____ ____ ____ ____ ____ ____ 25. 26. 27. 28. 29. 30. 31. 32. 33. 34. ____ ____ ____ ____ 35. 36. 37. 38. Which particles in atoms have a negative electric charge? What information in the periodic table indicates the number of protons in an atom? Most metals are NOT In the periodic table, the most reactive metals are found The elements that do not ordinarily form compounds are Fluorine, chlorine, bromine, and iodine are part of a family called Electrons involved in bonding between atoms are If atoms of a halogen nonmetal (Group 17) gain one electron, the atoms then have When an atom loses an electron, it becomes a An ionic bond is the attraction between A chemical bond formed when two atoms share electrons is called a(n) Molecular compounds do not conduct electricity because they The only sure evidence for a chemical reaction is A chemical reaction that absorbs energy in the form of heat is described as The substances listed on the left side of a chemical equation are the In chemical reactions, what does the principle of conservation of mass mean? Which of the following is a balanced chemical equation? In a chemical equation, numbers often appear in front of a chemical formula. These numbers tell you the What type of reaction is FeS + 2 HCl FeCl2 + H2S? The minimum amount of energy that has to be added to start a reaction is the A liquid mixture in which particles can be seen and easily separated by settling or filtration is a How is a solute different from a solvent in a solution? When a few spoonfuls of sugar are mixed into a cup of water, sugar is the A mixture containing particles that are too small to be seen easily but are large enough to scatter a light beam is called a How would a solute affect the boiling point of water? When a solute is added to a solvent, the freezing point of the solution is Weak tea is an example of a When you add so much solute that no more dissolves, you have a Which is a characteristic property of acids? Which is a likely use for a base? In a water solution, how do acids differ from bases? In water, bases form The pH scale measures If you have a solution of a strong acid and a solution of a weak acid of equal concentration and volume, then the Normal rainfall is slightly acidic, which means its pH must be You can find the pH of a substance by using Neutralization is a reaction between a(n) What does a neutralization reaction produce? 39. Describe the differences among solutions, colloids, and suspensions. 40. You are given three clear, colorless, unknown solutions, and red and blue litmus papers. Describe the steps you might take to correctly identify each solution as acidic, basic, or neutral. 41. Describe how solutions of acids and bases differ in terms of particles. How do the differences relate to the properties of acids and bases?