Key
advertisement

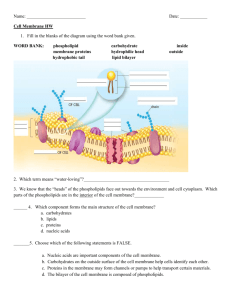
Bio102 Problems Fluid Mosaic Model 1. A peptide bond is formed by a(n) ___________. A. dehydration reaction B. equilibrium reaction C. hydrogen bond D. hydrolysis reaction E. disulfide bond 2. Which one of the following is an example of a secondary structure? A. Beta Sheet B. Triglyceride C. Double Helix D. Disulfide Bridge E. Peptide Bond 3. Which one statement accurately describes condensation reactions? A. Condensation reactions are used to break down polymers into monomers. B. Water is consumed during condensation reactions. C. A condensation reaction is the same thing as a hydrolysis reaction. D. Condensation reactions can be used to connect amino acids with peptide bonds. E. Condensation reactions are used to connect phospholipids together to make a cell membrane less permeable. 4. Which one force does NOT contribute to the tertiary structure of a protein? A. Hydrogen bonding B. Micelle interactions C. van der Waals interactions D. Disulfide bridges E. Ionic interactions 5. Which one of the following forces does NOT contribute to tertiary structure? A. Hydrogen bonds B. Amphipathic bonds C. Disulfide bonds D. Van der Waals (hydrophobic) interactions E. Ionic bonds 6. Which one of the following does NOT contribute to the secondary structure of a protein? A. Hydrogen bonding B. Activation energy interactions C. Hydrophobic interactions D. Ionic bonding E. Disulfide bridges 7. Not all proteins have quaternary structure. This is because not all proteins A. contain peptide bonds. B. have quaternary amines. C. contain at least one disulfide bridge. D. contain the amino acid quaternine. E. bind another protein. 8. The fluid mosaic model of membrane structure predicts that the plasma membrane A. prevents the destruction of the cell by osmosis. B. is more fluid than the cell membrane. C. restricts the lateral movement of phospholipids. D. forms a rigid structure to prevent the loss of important molecules. E. contains proteins. 9. Which of the following might have a secondary structure? A. Phospholipid B. Cholesterol C. Amino Acid D. Tyrosinase E. Peptide Bond 10. If a protein is amphipathic it is likely A. to be made of many amino acids. B. found in a membrane. C. to have quaternary structure. D. to be an enzyme. E. to be at or near it’s Vmax value. 11. Protein fill-in-the-blank: A. The “back” end of a protein is better called the ___Carboxy terminus_______. B. Individual units are held together by this special type of covalent bond: __Peptide bond__. C. A protein is a polymer of many __amino acids__________. D. An -helix is one type of ___secondary structure_. 12. We’ve drawn biological membranes as two layers of phospholipid molecules (shown as a circle with two tails) with large protein blobs (shown below as a striped ‘T’). Protein This membrane has an aqueous (or water-based) solution on both sides. Imagine that we took this membrane and instead of having it in water, we placed a hydrophobic liquid on both sides. How would the membrane structure change? Include both phospholipids and the protein shown above in your drawing. 13 Show the chemical structures of the three groups attached to the central carbon of an amino acid. H H H-N- O C -C-OH Side Group 13B The ‘front’ end of a protein is better known as the _ Amino- (or N-) terminus __. 13C An -helix (alpha-helix) is an example of a type of __Secondary_________ structure. 13D. Two amino acids are joined together with what type of bond? ___Peptide Bond_______ 13E. On what cellular structure are these bonds synthesized? _____Ribosome_______ 14. In a fully functional hemoglobin molecule, four individual proteins are associated with one another. If we increase the sodium concentration to 0.5M, the four proteins separate into individual proteins. Which type of structure has been disrupted? A. Primary B. Secondary C. Tertiary D. Quaternary 15. Each of the following four forces helps to stabilize the secondary and tertiary structure of a protein. Which one is the strongest force? A. Ionic interaction B. Disulfide bond C. Hydrophobic interaction D. Hydrogen bond 16. The image on the right is Figure 7.2 from your textbook, with some additional labels added. 16A. Would the G value for moving the circled phospholipid from its current position to “position A” be positive, negative or zero? Why? Zero. The phospholipid is equally stable (and thus has the same level of free energy) at either position 16B. Would the G value for moving the circled phospholipid from the innerlayer (position A) to the outer layer (position B) of the membrane be positive, negative or zero? Why? Zero. The phospholipid is equally stable (and thus has the same level of free energy) at position. B A either 16C. Which of these two movements happens faster? Why? The movement to “A” happens faster because it has a much lower activation energy 17. Using a very clever labeling technique, cell biologists can covalently modify the proteins in one region of a cell’s plasma membrane. Shown on the right is a human white blood cell. Suppose that we use this technique to ‘label’ all the plasma membrane proteins in the circle shown on the diagram. Those proteins now glow red under a special type of microscope. Immediately after labeling, only the circle glows bright red. Five minutes after labeling, all of the left half of the cell glows a dim red. Thirty minutes after labeling, the entire cell glows pink. 17A. Please briefly explain why the red glow slowly spreads across the surface of the cell. The red-labeled proteins diffuse away from this spot as predicted by the fluid mosaic model 17B. The major fatty acid found in the phospholipids of this cell is oleic acid, which is 18 carbon atoms long and has one unsaturation. If the major fatty acid was instead linoleic acid (18 carbon atoms, two unsaturations), how would this have changed the outcome of the experiment? Specifically, what do you think the cell would minutes after labeling one spot? Please explain your reasoning. look like five The red label would diffuse away from the one spot more rapidly. Thus, after five minutes the entire cell may be pink 18A. Shown below are the structures of three amino acids. Draw the structure of the small protein: N terminus – Leucine – Serine – Aspartic Acid – C terminus H O H O H | || | || | H2N—C—C—NH—C—C—NH—C—COOH | | | CH2 CH2OH CH2COOH | CH3-CH-CH3 Aspartic Acid Leucine Serine 18B. Which of these three amino acids is most hydrophobic? ____Leucine___ 19. Which of the following molecules is an amino acid? Circle all that apply. 20. A region of a protein that contains lots of amino acids with hydrophobic side chains is likely… A. a -sheet. B. a transmembrane domain. C. an allosteric site. D. a triacylglyceride. E. an active site. 21. In the lab, we’ve been studying the Fluid Mosaic Model by using living yeast cells. Using some sophisticated instruments, we determine that integral membrane proteins move at a rate of 10m/second at room temperature (25oC). 21A. What would happen to the rate of movement of this integral membrane protein if we increased the the temperature to 37oC? Please briefly explain your answer. It’s rate of lateral movement would increase because at higher temperatures the membrane becomes more fluid 21B. -sitosterol is a molecule made by most plants and it’s structure is shown on the right. If we simply add sitosterol to the liquid around the yeast cells, it becomes part of the cell membrane. Why doesn’t it stay in the liquid outside of the cells? Nearly all of this molecule is highly hydrophobic and thus very unstable in the aqueous liquid outside of the cells. It is much more stable when dissolved in the hydrophobic zone of the cell membrane. 21C. What effect will the addition of -sitosterol to the membrane have on the rate of movement of the integral membrane proteins at room temperature? Please briefly explain your answer. It should decrease it. -sitosterol is a sterol (like cholesterol) and thus it decreases the fluidity of membranes. 21D. We can also measure the rate at which ethanol (CH3-CH2-OH) crosses the cell membrane. Will adding -sitosterol increase or decrease the rate at which ethanol moves? Why? It will decrease ethanol’s movement across the membrane since decreases in fluidity are always associated with decreases in permeability. 21E. Name two modifications of the fatty acids present in the phospholipids that would decrease the rate at which the integral membrane proteins moves. For both modifications, what effect would this have on the rate of movement of ethanol across the membrane? You do not need to explain your reasoning for this answer. Modification Effect on ethanol movement rate longer fatty acids decrease more saturated fatty acids decrease 22. The three dimensional structure of a functional hemoglobin protein is shown below. 22A. Four different proteins are held together in this structure. Because of that, we know that this protein has __quaternary____ structure. 22B. Name the four types of molecular forces that may contribute to holding these proteins together. Disulfide bonds Ionic interactions Hydrogen bonds Hydrophobic interactions 22C. Which one of the four forces from 22B is a covalent bond? Disufide bons 22D. The structure of hemoglobin contains many spiral or spring-like structures that I’ve magnified and isolated below the big structure. What is the name of this structure? It’s an example of what level of protein structure? -helix. Secondary structure 22E. The ‘back’ end of a protein is better known as the ______Carboxyl terminus_______.