Conversion Factors
advertisement

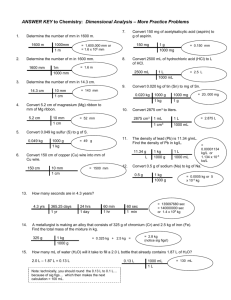
Name: ________________________ Hour: ____ Date: ___________ Chemistry: Conversion Factors Below are some conversion factors used in the SI System, and which we will use in this class. kilo- = 1000 1 kg = 1000 g 1 km = 1000 m 1 kL = 1000 L centi- = 1/100 milli- = 1/1000 Other Conversions 100 cm = 1 m 1000 mg = 1 g 1000 mm = 1 m 1000 mL = 1 L 1 mL = 1 cm3 1 L = 1 dm3 1 cm = 10 mm Solve each of the following problems. Show the correct set-up and always use units. 1. Determine the number of mm in 1600 m. 2. Determine the number of m in 1600 mm. 3. Determine the number of mm in 14.3 cm. 4. How many seconds are in 4.3 years? 5. Convert 2875 cm 3 to liters. 6. The density of lead (Pb) is 11.34 g/cm3. Find the density of Pb in kg/dm 3. 7. Convert 5.2 cm of magnesium (Mg) ribbon to mm of Mg ribbon. 8. Convert 0.049 kg sulfur (S) to g of S. 9. Convert 0.020 kg of tin (Sn) to mg of Sn. 10. Convert 150 mg of acetylsalicylic acid (aspirin) to g of aspirin. 11. Convert 2500 mL of hydrochloric acid (HCl) to L of HCl. 12. A metallurgist is making an alloy that consists of 325 g of chromium (Cr) and 2.5 kg of iron (Fe). Find the total mass of the mixture in kg. 13. How many mL of water (H2O) will it take to fill a 2 L bottle that already contains 1.87 L of H 2O? 14. Convert 150 cm of copper (Cu) wire into mm of Cu wire. 15. Convert 0.5 g of sodium (Na) to kg of Na. KEY Chemistry: Conversion Factors Below are some conversion factors used in the SI System, and which we will use in this class. kilo- = 1000 1 kg = 1000 g 1 km = 1000 m 1 kL = 1000 L centi- = 1/100 milli- = 1/1000 Other Conversions 100 cm = 1 m 1000 mg = 1 g 1000 mm = 1 m 1000 mL = 1 L 1 mL = 1 cm3 1 L = 1 dm3 1 cm = 10 mm Solve each of the following problems. Show the correct set-up and always use units. 1. Determine the number of mm in 1600 m. 100 cm 10 mm 6 x mm 1600 m 1600000 mm or 1.6 10 mm 1 m 1 cm 2. Determine the number of m in 1600 mm. 1 cm 1 m x m 1600 mm 1.6 m 10 mm 100 cm 3. Determine the number of mm in 14.3 cm. 10 mm x mm 14.3 cm 143 mm 1 cm 5. How many seconds are in 4.3 years? 365 days 24 hours 60 min 60 sec 8 x sec 4.3 years 1.36 10 seconds 1 year 1 day 1hour 1min 5. Convert 2875 cm 3 to liters. 1 mL 1L x L 2875 cm 3 2.875 L 3 1 cm 1000 mL 6. The density of lead (Pb) is 11.34 g/cm 3. Find the density of Pb in kg/dm 3. x kg g 11.34 3 dm cm3 1kg 1 cm3 1000 mL 1L kg 11.34 3 dm3 1000 g 1mL 1L 1 dm 7. Convert 5.2 cm of magnesium (Mg) ribbon to mm of Mg ribbon. 10 mm Mg 52 mm Mg ribbon x mm Mg 5.2 cm Mg 1 cm Mg 8. Convert 0.049 kg sulfur (S) to g of S. 1000 g S 49 g S x g S 0.049 kg S 1 kg S 9. Convert 0.020 kg of tin (Sn) to mg of Sn. 1000 g Sn 1000 mg Sn 20,000 mg Sn or 2.0 10 4 mg Sn x mg Sn 0.020 kg Sn 1kg Sn 1 g Sn 10. Convert 150 mg of acetylsalicylic acid (aspirin) to g of aspirin. 1 g aspirin 0.15 g aspirin (acetylsal icylic acid) x g aspirin 150 mg aspirin 1000 mg aspirin 11. Convert 2500 mL of hydrochloric acid (HCl) to L of HCl. 1L HCl x L HCl 2500 mL HCl 2.5 L HCl 1000 mL HCl 12. A metallurgist is making an alloy that consists of 325 g of chromium (Cr) and 2.5 kg of iron (Fe). Find the total mass of the mixture in kg. 1 kg Cr 0.325 kg Cr x kg Cr 325 g Cr 1000 g Cr Total 0.325 kg Cr 2.5 kg Fe 2.825 kg alloy 13. How many mL of water (H2O) will it take to fill a 2 L bottle that already contains 1.87 L of H2O? 2.00 L bottle - 1.87 L liquid 0.13 L empty 1000 mL H2 O 130 mL H2 O x mL H2 O 0.13 L H2 O 1L H2 O 14. Convert 150 cm of copper (Cu) wire into mm of Cu wire. 10 mm Cu 3 x mm Cu 150 cm Cu 1500 mm Cu or 1.50 10 mm Cu 1 cm Cu 15. Convert 0.5 g of sodium (Na) to kg of Na. 1kg Na 0.0005 kg Na or 5.0 10 -4 kg Na x kg Na 0.5 g Na 1000 g Na