carbonyl derivative
advertisement

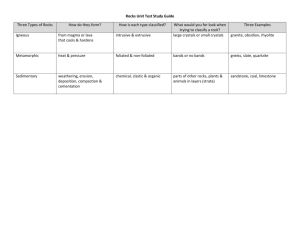
Experiment 3 Identification of a Carbonyl Compound by Preparing its Derivative Introduction Crystalline derivatives of many carbonyl compounds can be formed by condensation reactions with compounds such as phenylhydrazine and 2,4dinitrophenylhydrazine. These derivatives can usually be isolated in relatively pure forms which have well defined melting points. Phenylhydrazine forms derivatives (phenylhydrazones) readily with aromatic aldehydes, but in general 2,4dinitrophenylhydrazine is preferred because its derivatives (2,4dinitrophenyihydrazones) have higher melting points and are less soluble. These derivatives are useful in identification of carbonyl compounds. Chemicals 2,4dinitrophenylhydrazine in methanol, ethanol, methanol, carbonyl compound labelled X (different groups of students may work on different carbonyl compounds), dilute H2S04 Apparatus Beaker, 100 cm3 measuring cylinder, apparatus for melting point determination, apparatus for suction filtration, rubber rings (cut from a rubber tubing of appropriate diameter), ice bath Procedure [Hazard Warning: Methanol solution of 2,4dinitrophenylhydrazine is flammable and toxic, methanol, ethanol and many carbonyl compounds are flammable, and bench dilute sulphuric(V1) acid is corrosive.] A. Preparation of 2,4dinitrophenylhydrazone of compound X 1. Add 10 drops of compound X to 5 cm3 of 2,4dinitrophenyihydrazine solution in a 50 cm3 beaker. If crystals are not formed, add about 1 cm3 of dilute sulphuric(V1) acid. If they are still not formed, warm the mixture gently, then cool with scratching in ice water. 2. Filter off the crystals by suction filtration. While still on suction, wash the crystals with 1 cm3 of methanol. 3. Recrystallize the crystals from hot ethanol as follows: (a) Transfer the crystals to a 100 cm3 beaker standing on a steam bath (or in a 250 cm3 beaker of hot water). (b) Dissolve the crystals in the minimum amount of hot ethanol. (c) When the crystals have dissolved, cool the solution in an ice-water mixture until crystals reappear. (d) Filter the crystals by suction. If necessary, rinse the beaker with the filtrate (not the extra solvent) to complete the transfer. Finally, wash the crystals with a few drops of cold ethanol. Dry the crystals by drawing air through them for a few minutes. 4. Spread the crystals on a dry watch glass and leave overnight in a desiccator for drying. B. Determination of the melting point of the 2,4dinitrophenylhydrazone of compound X 1. Introduce a small amount of the crystals into a melting point tube until a total length of about 0.5 cm is compacted at the bottom of the tube. 2. Attach the prepared melting point tube to the thermometer, as shown in Figure 1. 3. Half-fill a boiling tube with paraffin oil, and position the thermometer with attached tube and the stirrer as shown in Figure 2. 4. Position the apparatus over a low Bunsen flame and gauze and gently heat the apparatus, stirring the paraffin oil all the time by moving the stirrer up and down. 5. Note the temperatures when the crystals start to melt and when the melting is completed. Figure 1 Figure 2 Carbonyl derivative / page 1 6. Compare the melting point of the crystals with the values given in the following table and thus identify compound X. Name Formula Melting point of Boiling point/C 2,4-dinitrophenylhydrazone/C Aldehydes methanal HCHO 167 21 ethanal CH3CHO 146, 164 (2 forms) 21 propanal CH3CH2CHO 156 48 butanal CH3CH2CH2CHO 123 75 (CH3) 2CHCHO 187 2methylpropanal 64 C6H5CHO 237 benzaldehyde 178 Ketones propanone CH3COCH3 56 128 butanone CH3CH2COCH3 80 115 pentan-2-one CH3CH2CH2COCH3 102 141 pentan-3-one CH3CH2COCH2CH3 102 156 hexan-2-one CH3CH2CH2CH2CO2CH3 128 107 (CH ) CHCH COCH 117 95 4-methylpentan2one 3 2 2 3 cyclohexanone 156 162 Discussion 1. What soluble impurities may be present in your product before recrystallization? How can they be removed in the recrystallization process? 2. What factors should be considered in selecting a suitable solvent in the recrystallization step? 3. In the recrystallization procedure, why were the crystals dissolved in only the minimum amount of hot ethanol? 4. If the sample contains insoluble impurities such as pieces of filter paper, cork, etc., suggest how they can be removed. 5. If the melting point of the 2,4dinitrophenylhydrazone is 156C, suggest how you can confirm whether compound X is propanal or pentan3one. Carbonyl derivative / page 2