02bonds
advertisement

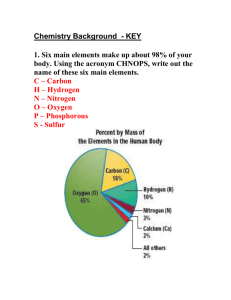
INTRODUCTION TO CHEMISTRY I. CHEMICAL BONDS A. Full outer shell rules 1. Atoms will react to fill their outermost shell a) Hydrogen and Helium only have one shell, which is filled with 2 electrons b) Other atoms have two or more shells, the outermost of which is filled with 8 electrons B. Ionic reactions 1. Definition a) Atoms are bonded to each other via opposite charges b) Charges result from gain or loss of electron to fill the atom's outermost shell (1) The metal atoms loses the electron(s) and non-metal atoms gain electron(s) 2. Example 1 a) Na has 1 electron in its outer shell and Cl has 7 electrons in its outer shell (1) Na loses one electron (a) Now its new outer shell is full with 8 electrons (2) Cl gains one electron (a) Now its outer shell is filled with 8 electrons b) Sodium now has a positive charge chlorine has a negative charge (1) There is an attraction between sodiums positive charge and chlorines negative charge 3. Example 2 a) Mg has 2 electrons in its outer shell and Cl has 7 electrons in its outer shell (1) Magnesium will lose both outer shell electrons (a) Now its new outer shell is full with 8 electrons (2) Two chlorine atoms can each accept one of electron from Mg (a) Now their outer shells are filled with 8 electrons C. Covalent reactions 1. Definition a) Atoms fill their outermost shell by sharing electrons 2. Single covalent bond a) Hydrogen atoms have only 1 electron in their outermost shell (1) To be stable, they must gain an electron to fill their outermost shell (2) Two hydrogen atoms will their electrons, thus filling both atoms outermost shell 3. Double covalent bond a) Oxygen has 8 electrons (1) To fill their outermost shell, two oxygen atoms will share 2 electrons each with the other oxygen (2) The oxygen atoms share two pairs of electrons 4. Triple covalent bond a) Nitrogen atoms, with 5 electrons, will share three pairs of electrons to fill their outermost shell D. Hydrogen bonds 1. Definition a) Electronegativity (1) Affinity of an atom for electrons (2) Different atoms have different electronegativities 2. Characteristics a) Oxygen and nitrogen are more electronegative than hydrogen (1) When hydrogen is covalently bound to O or N, the hydrogen will have a partial positive charge (2) It will therefore be attracted to atoms with slightly negative charges, usually another O or N E. Bonds in proteins and nucleic acids 1. Covalent bonds hold polymer together 2. Hydrogen and ionic bonds give molecule its three-dimensional structure II. ORGANIC COMPOUNDS A. Definition 1. Organic compound a) Compounds containing a carbon atom attached to a hydrogen atom b) Compounds that do not carbon with attached hydrogen are inorganic molecules 2. Hydrocarbons a) An organic compound whose molecules consist entirely of carbon and hydrogen atoms 3. Aliphatic a) Hydrocarbons and their oxygen or nitrogen derivatives that are not aromatic are called aliphatic compounds 4. Cyclic a) Aliphatic compound with a ring structure 5. Aromatic a) Benzene or contains benzene ring B. Bond formation 1. Carbon has 4 electrons in its outer shell a) Therefore it will form 4 covalent bonds C. Functional groups 1. Hydroxyl a) Oxygen and hydrogen covalently bonded b) - OH 2. Carboxyl a) A carbon atom with a double bonded oxygen and hydroxyl group b) - COOH 3. Amino a) Nitrogen with two covalently attached hydrogens b) - NH2 4. Sulfhydryl a) Sulfur with two covalently attached hydrogens b) - SH2 D. Types of organic molecules 1. Alcohols a) An molecule with a hydroxyl group attached to a carbon b) -R-CH2OH 2. Aldehydes a) An molecule with a double-bounded oxygen attached to a terminal carbon b) -R-CH2O 3. Organic acids a) An molecule with a double-bonded oxygen and a hydroxyl group attached to a carbon b) -R-COOH 4. Amine a) An molecule with an amine group attached to a carbon b) -R-CNH2 5. Ketone a) An molecule with a double-bonded oxygen attached to a carbon attached to two other carbons b) -R-CRO