Enquiry Reference: 13
advertisement

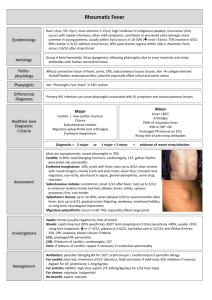
Faculty of Sexual and Reproductive Healthcare Clinical Effectiveness Unit A unit funded by the FSRH and supported by NHS Greater Glasgow and Clyde MEMBER’S ENQUIRY RESPONSE Enquiry Reference: 4464 Sent: 1.10.14 Prepared: 17.09.14 A: Question Can women with Sydenham’s chorea use hormonal methods of contraception? B: Response Sydenham’s chorea (rheumatic chorea) is the most common manifestation of rheumatic fever and of acquired chorea in the young. The introduction and use of antibiotics during the twentieth century resulted in a decline in the number of reported cases, especially in the developed world. However, in recent years a rise in the number of cases has been observed and this is thought to be associated with group A beta haemolytic streptococcal infection. Sydenham’s chorea is characterised by involuntary movement, hypotonia, and mild muscle weakness. Chorea can be generalised or unilateral and usually involves the face, hands, and arms. Sydenham’s chorea may also present with accompanying psychiatric disorders, for example, obsessive compulsive disorder (OCD) and anxiety. The summary of product characteristics (SPC) for Microgynon® lists the exacerbation of chorea as an adverse event reported post marketing. The British National Formulary (BNF) lists chorea as a contraindication for combined hormonal contraception (CHC). A search of the professional literature identified a case report 1 which reported a case of Sydenham’s chorea in association with combined oral contraceptive use in a healthy woman with no family history of the condition. The study also included a literature review and reported that 50 such cases have been reported in the professional literature.1 The study reported that in the majority of cases patients reported a past history of basal ganglia abnormalities, including Sydenham’s chorea. However, the study also reported that cases of contraceptive-induced chorea have also been reported.1 Another case report2 stated that the use of combined oral contraceptives is an uncommon but recognised risk factor for chorea. The study hypothesised that the underlying mechanism of this relationship is unclear but that it is thought to be a reactivation of Sydenham’s chorea. However, the study also suggested that in some patients the underlying mechanism may be related to the development of an autoimmune disorder, for example, Systematic Lupus Erythematosus (SLE).2 No evidence was identified which examined the use of progetogen-only or non-hormonal methods of contraception. C: Evidence-Based Medicine Question (which guided our literature search strategy) Population: Women with Syndenham’s chorea Intervention: Hormonal methods contraception Outcome: Safety / appropriateness D: Information Sources The CEU searched the following sources in developing this Member’s Enquiry Response Source Searched Existing FSRH and RCOG guidance The National Guidelines Clearing House The United Kingdom Medical Eligibility Criteria for Contraceptive Use (2009) The United Kingdom Selected Practice Recommendations for Contraceptive Use (2002) The World Health Organization Medical Eligibility Criteria for Contraceptive Use (2009) The World Health Organization Selected Practice Recommendations for Contraceptive Use (2008) The Cochrane Library MEDLINE and EMBASE from 1996 to 2014 Information Identified See below No relevant information No relevant information No relevant information See below E: Evidence Reviewed Sydenham’s chorea (rheumatic chorea) is the most common manifestation of rheumatic fever and of acquired chorea in the young.3 The introduction and use of antibiotics during the twentieth century resulted in a decline in the number of reported cases, especially in the developed world. However, in recent years a rise in the number of cases has been observed and this is thought to be associated with group A beta haemolytic streptococcal infection.3 Sydenham’s chorea is characterised by involuntary movement, hypotonia, and mild muscle weakness. Chorea can be generalised or unilateral and usually involves the face, hands, and arms. 3 Sydenham’s chorea may also present with accompanying psychiatric disorders, for example, obsessive compulsive disorder (OCD) and anxiety. The condition is frequently self-limiting and commonly resolves spontaneously in between three to six months and rarely last longer than one year. Treatment is usually reserved for patients with Sydenham’s chorea severe enough to impede function. Common treatment options include anticonvulsants, steroids, and dopaminergic blockers. Less frequently immunologic treatment may be used, however this may be associated with significant side effects. Children with Sydenham’s chorea require prophylaxis against streptococcal infections until the age of 18.3 The United Kingdom Medical Eligibility Criteria for Contraceptive Use (UKMEC) 4 does not contain any recommendations regarding Sydenham’s chorea and hormonal methods of contraception. The summary of product characteristics (SPC) for Microgynon® lists the exacerbation of chorea as an adverse event reported post marketing.5 The British National Formulary (BNF)6 lists chorea as a contraindication for combined hormonal contraception (CHC). A search of the professional literature identified a case report1 which reported a case of Sydenham’s chorea in association with combined oral contraceptive use in a healthy woman with no family history of the condition. The study also included a literature review and reported that 50 such cases have been reported in the professional literature.1 The study reported that in the majority of cases patients reported a past history of basal ganglia abnormalities, including Sydenham’s chorea. However, the study also reported that cases of contraceptive-induced chorea have also been reported.1 Another case report2 stated that the use of combined oral contraceptives is an uncommon but recognised risk factor for chorea. The study hypothesised that the underlying mechanism of this relationship is unclear but that it is thought to be a reactivation of Sydenham’s chorea. However, the study also suggested that in some patients the underlying mechanism may be related to the development of an autoimmune disorder, for example, Systematic Lupus Erythematosus (SLE).2 No evidence was identified which examined the use of progetogen-only or non-hormonal methods of contraception. F: References (1) Vela, L., Sfakianakis, G. N., Heros, D., Koller, W., and Singer, C. Chorea and contraceptives: case report with pet study and review of the literature. Mov Disord. 2004; 19(3):349-352. (2) Miranda, M., Cardoso, F., Giovannoni, G., and Church, A. Oral contraceptive induced chorea: another condition associated with anti-basal ganglia antibodies. J.Neurol.Neurosurg.Psychiatry 2004; 75(2):327328. (3) Robertson W. Chorea in children. Washington DC, USA: Medscape Reference; 2014. Available at: http://emedicine.medscape.com/article/1181993-overview (Accessed: 17/09/14) (4) Faculty of Sexual and Reproductive Health Care. UK Medical Eligibility Criteria for Contraceptive Use. (UKMEC 2009). Available at: http://www.fsrh.org/admin/uploads/UKMEC2009.pdf. (Accessed: 17/09/14) (5) Bayer plc. Microgynon 30 Summary of Product Characteristics. Available at: http://www.medicines.org.uk/EMC/medicine/1827/SPC/Microgynon+30/#UNDESIRABLE_EFFECTS (Accessed: 17/09/14) (6) Joint Formulary Committee.British National Formulary. British National Formulary: BNF 67. British Medical Association and Royal Pharmaceutical Society; 2014. The advice given in this Member's Enquiry Response has been prepared by the FSRH Clinical Effectiveness Unit team. It is based on a structured search and review of published evidence available at the date of preparation. The advice given here should be considered as guidance only. Adherence to it will not ensure a successful outcome in every case and it may not include all acceptable methods of care aimed at the same results. This response has been prepared as a service to FSRH members, but is not an official Faculty guidance product; Faculty guidance is produced by a different and lengthier process. It is not intended to be construed or to serve as a standard of medical care. Such standards are determined on the basis of all clinical data available for an individual case and are subject to change as scientific knowledge advances. Members are welcome to reproduce this response by photocopying or other means, in order to share the information with colleagues. Enquiry response by JS Checked by LM