Formal Lab - Sites at Penn State

A Comparative Analysis of the Hardness of Various Water
Sources
Brandon Mentley
April 3, 2012
Chem 111, Section 103
Group Members: Ashley Mason, Kristen Mazzei, Cameron McClain, Todd
McMillen, Jake McTavish
TA: Bill Charette
Mentley 1
Introduction:
Water is perhaps the most important component for the survival of all life on Earth. In addition to drinking water, humans also need to use water for a variety of other purposes. For this reason, it is essential to have water of suitable quality. One aspect of this is water hardness.
Water hardness refers to the concentration of Ca
2+
, Mg
2+
, and other polyvalent cations in a sample of water. Hardness is generally expressed in units of milligrams of calcium carbonate per liter of water. Note that mg/L is equivalent to parts per million (ppm). It is most important to note the presence of calcium and magnesium since these two ions are by far the most prevalent in most water samples. A sample of water with a high concentration of these polyvalent cations is said to be “hard,” while a sample with a low concentration of these ions is said to be “soft.”
These ions can be found in soil and rocks, and they become dissolved in water as it flows. The hardness of water is important for a variety of reasons. A process tied to water hardness known as “scale formation” is a very common and troublesome problem in factories. Basically, boilers and evaporators used in industry evaporate water, and any minerals that were dissolved in the water will be left behind. This results in the formation of a hard layer that is known as scale.
This scale can lower the performance of machines because it clogs pipes and valves. Removing this scale is a hassle at best and can be a very large expense for companies at worst. Using relatively soft water can help to avoid these problems, hence the importance of knowing how to soften water
1
. Another problem with the use of hard water is that laundry and washing machines require more soap or detergent to be effective if hard water is used rather than soft water
2
. In addition, hard water is not ideal for bathing since it interacts with soap in such a way that it leaves a sticky film on the skin. However, water hardness is far from being entirely bad. The
National Research Council has found that drinking hard water is not hazardous to one’s health
Mentley 2 and can actually supplement dietary requirements for calcium and magnesium
3
. Additionally, a study conducted at the Dartmouth Medical School linked water hardness to lower risk of several diseases, including hypertension
4
.
There are several methods for the removal of the ions that contribute to water hardness.
As was seen in this lab, the addition of sodium carbonate (washing soda) is one method. The sodium carbonate reacts with calcium and magnesium to form insoluble precipitates. Calcium hydroxide (lime) can also be used in the same manner. Another method used in this lab is cation exchange resin. The resin contains either Na
+
or H
+
and is able to soften water by exchanging these monovalent cations for the divalent cations that cause water hardness. After the resin is used, it can be regenerated by rinsing the resin with an excess of NaCl, which causes the reverse reaction to occur, returning the resin to its original state
1
. One side-effect of using ion-exchange resin is that it results in the addition of sodium to the water that it softens. Excessive sodium intake can lead to health problems, such as hypertension
5
. If H
+
is exchanged instead of Na
+
, the result will be water that is more acidic. An additional method is the use of ethylenediaminetetraacetic acid (EDTA), which is also useful for measuring the hardness of water
1
.
There are several methods for determining the hardness of a sample of water. An important distinction to make is the difference between total dissolved solids (TDS), which consists of everything that remains in the residue after a sample of water is evaporated, and total divalent cation content, which only consists of the concentrations of divalent cations, most importantly Ca
2+
and Mg
2+
, in the sample. Perhaps the most important and useful method for measuring the hardness of water is the use of ethylenediaminetetraacetic acid (EDTA) in conjunction with eriochrome black T (EBT), which functions as an indicator. In order to use this
Mentley 3 method, the pH of the sample is first set at 10 using a buffer. This causes the EBT to appear blue. The presence of Mg 2+ causes the EBT to turn the color of red wine. Because this only happens in the presence of Mg
2+
, the sample in question must contain Mg
2+
in order for the indicator to be effective. When EDTA is added, it binds to divalent cations, most importantly
Ca
2+
, which has no effect on the color of the solution. It binds last to Mg
2+
. Once all of the Mg
2+ has reacted, the EBT will once again be blue, indicating that the titration is complete. The number of moles of EDTA used to complete the titration will correspond directly to the number of moles of divalent cations in the sample
1
.
Another method is Atomic Absorption Spectrophotometry (AA). In order to carry out an analysis by AA, the water sample must be aerosolized and sprayed into a flame that reaches
2300°C. At the same time, monochromatic light with a ΔE that corresponds to the ΔE of the atoms being analyzed (Ca 2+ or Mg 2+ ) is emitted by a hallow cathode lamp. This light will only be absorbed by atoms of the element in question. After this light has passed through the flame, it passes through a monochrometer, which only allows light of the specific wavelength being emitted by the lamp to pass through. This light then falls on a detector, and the spectrophotometer is able to give a reading for the absorbance (A) of the aerosolized sample.
Using the equation A = abc, one can easily determine the concentration of the sample in question. In this lab, a list of known absorbance and concentration readings was used to create an equation for the conversion from absorbance to concentration. This method allows us to ignore the variables “a” and “b” since they are the same in each case. The variable “c” refers to the concentration and is directly proportional to absorbance. Since AA only tests for the presence of one element at a time, it must be performed separately for Ca 2+ and Mg 2+ . AA can only accurately produce results if the Ca
2+
concentration is below 50 mg/L and the Mg
2+
Mentley 4 concentration is below 30 mg/L. If the concentrations exceed these values, the sample must be diluted before it can be properly analyzed. In contrast to the EDTA method, AA tests exclusively for Ca
2+
and Mg
2+
, while the EDTA titration accounts for all divalent cations
1
.
Water samples from a tap in a hotel in Atlantic City, New Jersey; a tap in Huntingdon,
Pennsylvania; a tap in Pinchot Hall in University Park, Pennsylvania; a tap in a hotel in Aruba; a tap on a farm in Perry County, Pennsylvania; and a creek in Audubon, Pennsylvania were analyzed in order to determine their hardness 6,7,8,9,10,11 . The United States Geological Survey
(USGS) defines water with hardness from 0-60 mg/L to be soft, water with hardness from 61-
120 mg/L to be moderately hard, water with hardness from 121-180 mg/L to be hard, and water with hardness exceeding 180 mg/L to be very hard. All of these values are expressed in terms of milligrams of calcium carbonate per liter of water
12
. It should be noted that the units that will be used in expressing hardness from this point forward are parts per million (ppm), which is equivalent to mg/L. Based on the map provided by the USGS, it is hypothesized that the samples from Huntingdon, Perry County, and Audubon will all be soft to moderately hard
10
. The water in State College is known to be very hard, so that is the expected result for the water from
Pinchot Hall
13
. Hotels tend to soften their water, so it is expected that the water samples from
Atlantic City and Aruba will be the softest samples tested 14 .
Procedure:
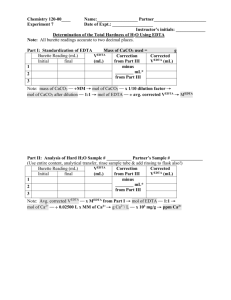
Water samples were taken from the previously mentioned sources using proper transfer technique. This means that the containers used to store the samples were rinsed with distilled water, rinsed with water from the source being sampled, and then filled with the sample. These samples were then analyzed using techniques outlined in the PSU Chemtrek. One technique used was a simple evaporation of the water samples in order to examine the residue left behind
Mentley 5 and do a qualitative analysis of the TDS by comparing the residue to that left by distilled water and 1 x 10 -3 M Ca 2+ . The hardness was also measured using AA. Measurements for the absorbance of samples with known concentrations of Ca
2+
and Mg
2+
were used in order to create a linear regression equation to convert from absorbance to concentration. This equation was used with the absorbance readings given by the AA in order to determine the concentrations of
Ca
2+
and Mg
2+
in the samples. These measurements were then converted to concentration in terms of CaCO
3
. The last technique used was an EDTA titration. One drop of each sample was added to each well of a 1 x 12 well along with a NH
3
/NH
4
Cl buffer and the EBT indicator. A serial titration was then performed by adding progressively larger numbers of drops of 2 x 10
-4
M
EDTA to each well. After observing how many drops were required in order for the EBT to change colors, the concentration of divalent cations could be determined since the moles of
EDTA used is equivalent to the moles of divalent cations. Drops could be used as a unit of volume since a standard drop size was used. The EDTA titration was also performed on each sample after it had been treated with washing soda and again after it had been treated with ionexchange resin in order to investigate the effectiveness of these softening agents
1
. The samples from Huntingdon, Pinchot Hall, and the creek in Audubon were diluted 1:1 with distilled water before all analyses were carried out in order to ensure an accurate reading by AA and to lower the volume of EDTA required to titrate the sample
7,8,11
. The sample from Perry County was also diluted 1:1, but only for the EDTA analysis and the AA analysis of Ca
2+ 10
.
Results:
In order to make use of the absorbance values determined through AA analysis, it was necessary to use known values for absorbance and concentration to create equations for converting between absorbance and concentration.
Mentley 6
Figure 1: Calibration Graph for Conversion from Absorbance to Ca 2+ Concentration
0,5
0,45
0,4
0,35
0,3
0,25
0,2
0,15
0,1
0,05
0
0
Absorbance vs. Ca
2+
Concentration
10 y = 0,0093x + 0,0112
20 30
Concentration (ppm)
40 50 60
Figure 2: Calibration Graph for Conversion from Absorbance to Mg 2+ Concentration
0,6
0,5
0,4
0,3
0,2
0,1
0
0
Absorbance vs. Mg
2+
Concentration
5 10 y = 0,0151x + 0,0245
15 20
Concentration (ppm)
25 30 35
In order to account for the dilution factor, the concentrations for any samples that were diluted
1:1 were multiplied by a factor of 2. The concentrations of Ca
2+
and Mg
2+
were converted to the concentration of CaCO
3
as this is the standard unit used to express hardness.
Pinchot Hall
Ca
2+
Mg
2+
Aruba
Ca
2+
Mg 2+
Perry Country
Ca
2+
Mg
2+
Audubon
Ca
2+
Mg
2+
Mentley 7
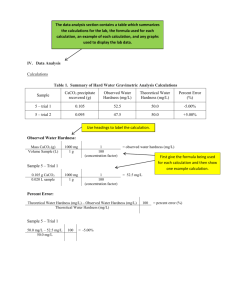
Table 1: Results of Atomic Absorption Spectrophotometry
Sample Absorbance (nm) Ion Concentration
(ppm)
Atlantic City
Ca
2+
Mg
2+
Huntingdon
Ca
2+
Mg 2+
0.0571
0.0363
0.1802
0.1454
4.94
0.781
36.34
16.01
Concentration (ppm of CaCO
3
)
15.6
156.8
339.8 0.3636
0.3000
0.0458
0.0210
0.3222
0.2412
0.2103
0.1455
75.78
36.49
3.72
-0.232*
66.88
14.35
42.82
16.03
9.30
226.3
173.1
Mentley 8
*The absorbance value for Mg
2+
for this sample was outside the range of values for which the equation could produce an accurate concentration. The concentration of Mg 2+ for this sample was considered to be zero for all further analyses.
The ion concentrations were determined using the linear regression equations displayed in Figure
1 and Figure 2.
Sample calculation for Ca
2+ concentration in Audubon sample: y = 0.0093x + 0.0112
0.2103 = 0.0093x + 0.0112 x = 21.41 ppm
21.41 ppm diluted * (2 ppm undiluted/1 ppm diluted) = 42.82 ppm Ca 2+
Sample calculation for Mg
2+ concentration in Audubon sample: y = 0.0151x + 0.0245
0.1455 = 0.0151x + 0.0245 x = 8.013 ppm
8.013 ppm diluted * (2 ppm undiluted/1 ppm diluted) = 16.03 ppm Mg 2+
Sample calculation for hardness in terms of CaCO
3
concentration for Audubon sample:
[CaCO
3
] = 42.82 ppm Ca
2+
* (2.5 ppm CaCO
3
/1 ppm Ca
2+
) + 16.03 ppm Mg
2+
* (4.12 ppm
CaCO
3
/1 ppm Mg
2+
) = 173.1 ppm CaCO
3
The conversion factors for converting from the concentrations of Ca 2+ of Mg 2+ to concentration of CaCO
3
were derived based on the ratio of the molar mass of CaCO
3
to that of the ions in question.
The hardness of each sample was also determined using an EDTA titration. The hardness was calculated based on the number of drops required for the EBT to change from a red wine color to blue.
Table 2: Results of EDTA Analysis
Sample
Atlantic City
Original Sample
Sample Softened with
Washing Soda
Sample Softened with Ion-
Exchange Resin
Huntingdon
Original Sample
Sample Softened with
Washing Soda
Sample Softened with Ion-
Exchange Resin
Pinchot Hall
Original Sample
Sample Softened with
Washing Soda
Sample Softened with Ion-
Exchange Resin
Aruba
Original Sample
Sample Softened with
Washing Soda
Sample Softened with Ion-
Exchange Resin
Perry County
Original Sample
Sample Softened with
Washing Soda
Sample Softened with Ion-
Exchange Resin
Audubon
Original Sample
Mentley 9
1
8
1
9/10*
2
6
4
1
Drops of EDTA Required for Color Change
3
3
1
1
6
5
8
7
20
320
40
18
40
160
40
240
Hardness (ppm of CaCO
3
)
60
60
20
240
200
40
320
280
Mentley 10
Sample Softened with
Washing Soda
Sample Softened with Ion-
Exchange Resin
4
1
160
40
*This sample was very soft, so 10 drops of the water sample were titrated with 9 drops of EDTA.
This vale is being written as 9/10 of a drop for ease of displaying it in the table.
Sample calculation of water hardness from EDTA analysis for Audubon sample:
[EDTA] * (V of EDTA) = [CaCO
3
] * (V of CaCO
3
)
(2 x 10
-4
M) * (6 drops) = [CaCO
3
] * (1 drop)
[CaCO
3
] = 1.2 x 10 -3 M
1.2 x 10
-3
M diluted * (2 M undiluted/1 M diluted) = 2.4 x 10
-3
M CaCO
3
2.4 x 10
-3
M CaCO
3
* (100000 ppm/1 M) = 240 ppm CaCO
3
Discussion:
Both the EDTA and AA analyses showed the taps in Pinchot Hall and on the farm in
Perry County to have the hardest water. They also showed that both samples taken from hotels were the softest. The relative hardness of the water from Perry County was not necessarily predicted in the hypothesis, but all other results confirm the hypothesis in regard to the relative hardness of the various samples. That is, it was predicted that the water from Pinchot Hall would be the hardest (it was; the AA value was 339.8 ppm); the Atlantic City and Aruba samples would be the softest, as they were; and the Huntingdon, Perry County, and Audubon samples would have comparable hardness, the exception to this being the hardness of the Perry County sample, which was higher than expected. While the hypothesis regarding the relative hardness was generally confirmed, the hardness of the water samples from Huntingdon, Perry County, and
Audubon was higher than had been predicted. These samples were expected to be rated as soft or moderately hard on the USGS’s scale 12
. However, their AA hardness values were 156.8 ppm,
226.3 ppm, and 173.1 ppm, respectively, which places them firmly in the range of hard to very hard. The USGS website does note that the hardness of water can vary quite a bit at a local level,
so its estimates for regional water hardness are not perfect
12
. The water samples from
Mentley 11
Huntingdon and Perry County likely flowed through pipes with large deposits of calcium and/or magnesium, especially since the Perry County sample came from a farm where pipe maintenance and replacement is probably not frequent. As for the Audubon sample, it can be assumed from the results that the rocks and soil over which the particular stream in question flows contain higher levels of calcium and magnesium than what is normal for the region, resulting in harder water. It is typical for many hotels to soften their water, so it is not surprising that the samples from Atlantic City and Aruba were the softest with AA results of 15.6 ppm and 9.30 ppm, respectively
14
. The sample from State College was expected to be very hard since State College is an area known for having very hard water, which was confirmed in this analysis
13
.
While this discussion has focused on the results produced by the AA analysis, the results produced by the EDTA titration support the same conclusions, despite some amount of difference between the two sets of results. It should be noted that there is a reason for the discrepancy between the hardness values obtained from the AA analysis and those obtained from the EDTA titration. First of all, due to the large jump in the concentration that is measured if the titration is off by a single drop, the EDTA analysis has a rather high margin of error compared to the AA. Also, EDTA tests for the presence of all divalent cations, while AA tests exclusively for
Ca
2+
and Mg
2+
. These two are by far the most prevalent divalent cations in water, but the presence of others may account for some of the differences between the values obtained from the two analyses.
There were a few sources of error inherent is this experiment. As was noted, a difference of a single drop in the EDTA titration could have a fairly large impact on the results. Any undiluted samples would have a margin of error of ±20 ppm. Samples diluted 1:1 would have a
Mentley 12 margin of error of ±40 ppm since the concentrations (and margins of error) for those samples needed to be multiplied by two in order to obtain the values for the undiluted samples. It is not unreasonable to assume that an error could be made here, especially since the point at which the
EBT has fully gone from wine-red to blue can be difficult to discern. Also, it is quite likely that the true concentration lies somewhere between the values corresponding to two different numbers of EDTA drops. A more precise measurement could have been obtained by titrating a larger volume of the water sample with a larger volume of EDTA. Also, drops were used as a unit of volume under the assumption that the drops were of uniform size, but it is quite likely that there was some variation in the size of the drops. Of course, human error is always a possible source of error. The dilutions may not have been entirely accurate, and it is possible that water samples could have been contaminated by outside sources.
The results show that the commercial softening agent (washing soda) was at least somewhat effective at softening the samples that began at a relatively high level of hardness. It was not, however, effective at softening the two samples taken from hotels. In fact, the sample from Aruba actually tested as harder than before being treated with the washing soda. The ionexchange resin proved very effective at softening all of the samples. The value recorded for the hardness of the Aruba sample after being treated with the resin was higher than the value for the untreated sample, but it seems unlikely that the sample actually became harder. This observation is more likely to be due to the fact that a much larger volume of the sample water was used for the titration of the untreated sample than for the treated sample, resulting in a more precise measurement for the untreated sample. Given the margin of error for the sample treated with ion-exchange resin, it is quite possible that this sample was as soft as or softer than the untreated sample.
Mentley 13
Conclusion:
The hypothesis was proven to be partially correct. As was expected, the samples from
Atlantic City (AA hardness value = 15.6 ppm) and Aruba (9.30 ppm) were the softest, while the sample from Pinchot Hall (339.8 ppm) was the hardest. It was not predicted that the sample from Perry County (226.3 ppm) would be significantly harder than the samples from Huntingdon
(156.8 ppm) and Audubon (173.1 ppm). Also, the samples from Huntingdon, Pinchot Hall,
Perry County, and Audubon were all harder than was expected. The EDTA results, while slightly different from the AA results, point to the same conclusion. The commercial softening agent was fairly effective, while the ion-exchange resin was extremely effective.
References:
1
Thompson, Stephen. PSU Chemtrek: Small-Scale Experiments for General Chemistry . 18 th
ed.
Ed. Joseph T. Keiser. Plymouth: Hayden-McNeil Publishing, 2012.
2 “Explanation of Water Hardness.”
Fairfax Water . <http://www.fcwa.org/water/hardness.htm>.
(accessed March 2012).
3 “Hard Water Problems.” <http://www.hardwater.org/hard_water_problems.html>. (accessed
March 2012).
4 Schroeder, Henry A. “Relations Between Hardness of Water and Death Rates From Certain
Chronic and Degenerative Diseases in the United States.” Journal of Chronic Diseases .
Volume 12, Issue 6. December 1960, pp. 586-591.
5
Brown, Theodore. Chemistry: The Central Science . 11 th
Edition. Upper Saddle River: Prentice
Hall, 2009, pp. 788.
6 Mazzei, Kristen, Chem 111 Laboratory Notebook, pp. 30-32.
7
Mason, Ashley, Chem 111 Laboratory Notebook, pp. 34-37.
8
McTavish, Jake, Chem 111 Laboratory Notebook, pp. 26-28.
9 McClain, Cameron, Chem 111 Laboratory Notebook, pp. 31-33.
10
McMillen, Todd, Chem 111 Laboratory Notebook, pp. 30-34.
11
Mentley, Brandon, Chem 111 Laboratory Notebook, pp. 19-21.
12 “Water Hardness and Alkalinity.”
United States Geological Survey .
<http://water.usgs.gov/owq/hardness-alkalinity.html#briggs>.
13 “State College Water May Cause Kidney Stones.” Voices of Central Pennsylvania .
<http://voicesweb.org/node/1670>.
14 “Water Softening… The Facts.”
Mentley 14
<http://www.watersoftenersaltuk.co.uk/pages/soft_water_benefits.php>.