Science - Cloudfront.net
advertisement
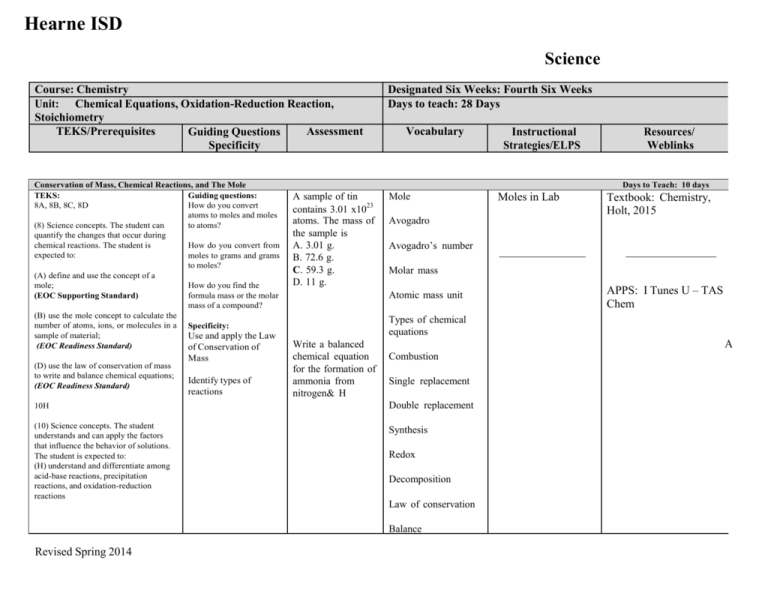
Hearne ISD Science Course: Chemistry Unit: Chemical Equations, Oxidation-Reduction Reaction, Stoichiometry TEKS/Prerequisites Assessment Guiding Questions Specificity Conservation of Mass, Chemical Reactions, and The Mole TEKS: Guiding questions: 8A, 8B, 8C, 8D How do you convert atoms to moles and moles (8) Science concepts. The student can to atoms? quantify the changes that occur during chemical reactions. The student is How do you convert from expected to: moles to grams and grams to moles? (A) define and use the concept of a mole; How do you find the formula mass or the molar (EOC Supporting Standard) mass of a compound? (B) use the mole concept to calculate the number of atoms, ions, or molecules in a Specificity: sample of material; Use and apply the Law (EOC Readiness Standard) of Conservation of (D) use the law of conservation of mass to write and balance chemical equations; (EOC Readiness Standard) Mass Identify types of reactions Designated Six Weeks: Fourth Six Weeks Days to teach: 28 Days Vocabulary Resources/ Weblinks Days to Teach: 10 days A sample of tin contains 3.01 x1023 atoms. The mass of the sample is A. 3.01 g. B. 72.6 g. C. 59.3 g. D. 11 g. Mole Moles in Lab Textbook: Chemistry, Holt, 2015 Avogadro Avogadro’s number Molar mass Atomic mass unit APPS: I Tunes U – TAS Chem Types of chemical equations Write a balanced chemical equation for the formation of ammonia from nitrogen& H A Combustion Single replacement 10H Double replacement (10) Science concepts. The student understands and can apply the factors that influence the behavior of solutions. The student is expected to: (H) understand and differentiate among acid-base reactions, precipitation reactions, and oxidation-reduction reactions Synthesis Redox Decomposition Law of conservation Balance Revised Spring 2014 Instructional Strategies/ELPS Hearne ISD Science Course: Chemistry Unit: Chemical Equations, Oxidation-Reduction Reaction, Stoichiometry TEKS/Prerequisites Assessment Guiding Questions Specificity Redox Chemistry TEKS: 10H (10) Science concepts. The student understands and can apply the factors that influence the behavior of solutions. The student is expected to: (H) understand and differentiate among acid-base reactions, precipitation reactions, and oxidation-reduction reactions (EOC Readiness Standard) College and Career Readiness Standards Revised Spring 2014 Vocabulary Instructional Strategies/ELPS Resources/ Weblinks Days to Teach: 5 Days Guiding Questions: Precipitate reaction Specificity: Understand and differentiate among acid-base reactions, precipitation reactions, and oxidation-reduction reactions Text: Modern Chemistry, Holt, 2015. Redox reactions How do you differentiate between precipitate and redox reactions? Oxidation number If zinc is placed in a copper sulfate solution, which of the following occurs? A. Zinc is oxidized B. Copper is the reducing agent C. Zinc is reduced D. Copper is oxidized E. Chemical reactions 1. Classify chemical reactions by type. Describe the evidence that a chemical reaction has occurred. 2. Describe the properties of acids and bases, and identify the products of a neutralization reaction. 3. Understand oxidation-reduction reactions. 5. Understand energy changes in chemical reactions. Designated Six Weeks: Fourth Six Weeks Days to teach: 28 Days In the reaction 2K + Br2 2K+ + 2Br–, which species is reduced? a. K only b. both K and Br2 c. Br2 only d. neither K nor Br2 Electroplating Lab Oxidize Reduce Redox Reactions: Lecture http://academicearth.org/l ctures/got-electrons-redox reactions Redox Reaction Demos: http://www.youtube.com/ atch?v=4-LA9BxQsdE Electroplating ELPS: http://ritter.tea.state.t x.us/rules/tac/chapter 074/ch074a.html 3E – QAR 4K – analyze lab data APPS: I Tunes U – TAS Chem A Hearne ISD Science Course: Chemistry Unit: Chemical Equations, Oxidation-Reduction Reaction, Stoichiometry TEKS/Prerequisites Assessment Guiding Questions Specificity Reaction Stoichiometry TEKS: 8E 8) Science concepts. The student can quantify the changes that occur during chemical reactions. The student is expected to: (E) perform stoichiometric calculations, including determination of mass relationships between reactants and products, calculation of limiting reagents, and percent yield. (EOC Supporting Standard) Prerequisites Dimensional analysis College and Career Readiness Standards G. The Mole and Stoichiometry 1.Understand the mole concept 2.Understand molar relationships in reactions, stoichiometric calculations, and percent yield Revised Spring 2014 Designated Six Weeks: Fourth Six Weeks Days to teach: 28 Days Vocabulary Instructional Strategies/ELPS Resources/ Weblinks Days to Teach: 15 Days Guiding Questions: What is the law of conservation of mass? Specificity: Use stoichiometry of chemical equations to calculate molar conversions • Calculate Mole ratio Limiting reactant • Calculate Percent Yield Determine the limiting reagent of a given chemical reaction. If 3 moles of water undergoes electrolysis, how many grams of oxygen and how many grams of hydrogen are produced? Chemical equations Products http://eocvideos.weebly.c m/ Reactants Percent yield Stoichiometry In a lab setting, 2 grams of potassium chlorate are decomposed into 1.15 grams of potassium chloride. What is its percent yield? Textbook: Modern Chemistry, Holt, 2015. Limiting reagent (reactant) Lab: The Decomposition of Potassium Chlorate ELPS: http://ritter.tea.state.t x.us/rules/tac/chapter 074/ch074a.html 4K – Q & A APPS: I Tunes U – TAS A Chem