Unit 6 Review Pre
advertisement

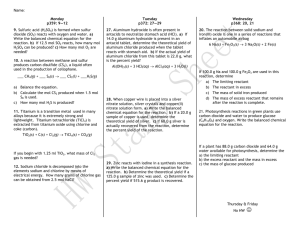
Unit 6 Review Chapter 9 - Stoichiometry Define the following key terms: Stoichiometry Balanced equation Law of Conservation of Mass Mole ratio Limiting reagents (reactant) Excess reactant Percent yield (equation) Actual yield Theoretical yield Mol to Mol calculations: 1) ____ NaCl ____ Na + ____ Cl2 How many moles of Cl2 can be produced from 2.50 mol NaCl? 2) 1 H2SO4 + 2 NaOH 1 Na2SO4 + 2 H2O How many mol Na2SO4 will be produced if 0.9 mol NaOH is completely reacted? Mass to Mass calculations: ____ K2CrO4 + _____ Pb(NO3)2 _____ PbCrO4 + _____ KNO3 3) How many grams of lead(II) chromate will be produced from 5.8 g of potassium chromate? 4) For the reaction: 2 Li + 1 Br2 2 LiBr How many grams of LiBr are produced if 45.95 g Br 2 react? Limiting reactants: 5) For the reaction: a. 2H2 + O2 2H2O If 8.5 moles of H2 react with 2.95 moles of O2, what is the limiting reactant? 6) For the reaction: a. 4 Li + O2 2 Li2O Given 7.45 moles of Li and 4.20 moles of O2, determine the limiting reactant 7) Sulfuric acid reacts with aluminum hydroxide by double replacement. For the reaction: H2SO4 + Al(OH)3 Al2(SO4)3 + H2O If 30.0 grams of sulfuric acid react with 25.0 g of aluminum hydroxide, identify the limiting reactant. Determine the mass of the Aluminum sulfate produced by the complete reaction of the limiting reactant. Determine the mass of the excess reactant remaining. Percent yield: 𝐩𝐞𝐫𝐜𝐞𝐧𝐭 𝐲𝐢𝐞𝐥𝐝 = 𝐚𝐜𝐭𝐮𝐚𝐥 𝐲𝐢𝐞𝐥𝐝 𝐱 𝟏𝟎𝟎 𝐭𝐡𝐞𝐨𝐫𝐞𝐭𝐢𝐜𝐚𝐥 𝐲𝐢𝐞𝐥𝐝 1. Calculate the percent yield for the following if the theoretical yield is 20.0 g with an actual yield of 15.0 g. 2. Calculate the actual yield if the theoretical yield is 2.3 g with a percent yield of 90.0%? 3. With the following reaction: C6H6 + Cl2 C6H5Cl + HCl When 36.8 g of C6H6 react with an excess of Cl2, the actual yield of C6H5Cl is 38.8 g. What is percent yield of C6H5Cl? (use mass-mass of limiting reactant to get determine the mass of C6H5Cl theoretical yield)