Limiting Reactant
advertisement

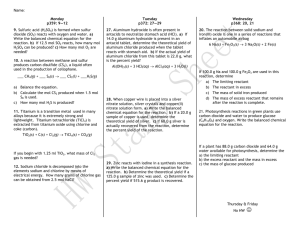
Section 9-3 Limiting Reactants and Percent Yield Example 1 You want to make smore’s according to the following recipe: 2 graham crackers + 1 marshmallow + 1 chocolate = 1 smore If you have 17 graham crackers, 7 marshmallows and 9 chocolates, which is the limiting ingredient? How many smore’s can you make? Limiting Reactant (or Reagent) The substance that is completely used up first in a reaction The reactant that limits the amounts of the other reactants that can combine and the amount of product that can form in a chemical reaction. Excess Reactant • The substance that is not used up completely in a reaction. Sample Problem 9-6 Silicon dioxide (quartz) is usually quite unreactive, but reacts readily with hydrogen fluoride according to the following equation: SiO2 (s) + 4 HF (g) → SiF4 (g) + 2 H2O (l) If 2.0 mol of HF are exposed to 4.5 mol of SiO2, which is the limiting reagent? Practice Problem #1 Some rocket engines use a mixture of hydrazine, N2H4, and hydrogen peroxide, H2O2, as the propellant. The reaction is N2H4 (l) + 2 H2O2 (l) → N2 (g) + 4 H2O (g) a. Which is the limiting reactant in this reaction when 0.750 mol of hydrazine is mixed with 5.00 mol of H2O2? b. How much of the excess reactant, in moles, remains? c. How much of each product, in moles, is formed? Practice Problem #2 If 20.5 g of chlorine is reacted with 20.5 g of sodium, which reactant is in excess? How do you know? Sample Problem 9-7 The black oxide of iron, Fe3O4, is the mineral magnetite. It can be made in the lab. 3 Fe (s) + 4 H2O (g) → Fe3O4 (s) + 4 H2 (g) a. When 36.0 g of steam is mixed with 167 g of Fe, which is the limiting reagent? b. What mass of iron oxide is produced? c. What mass of excess reagent remains when the reaction is completed? Practice Problem #1 Zinc and sulfur react to form zinc sulfide. 8 Zn (s) + S8 (s) → 8 ZnS (s) a. If 2.00 mol of Zn are heated with 1.00 mol of S8, identify the limiting reagent. b. How many moles of excess reagent remain? c. How many moles of the product are formed? Practice Problem #2 Carbon reacts with steam to produce hydrogen and carbon monoxide. a. If 2.40 mol of carbon are exposed to 3.10 mol of steam, identify the limiting reagent. b. How many moles of each product are formed? c. What mass of each product is formed? Theoretical Yield The maximum amount of product that can be produced from a given amount of reactant. Actual Yield The measured amount of a product obtained from a reaction. In most chemical reactions, the amount of product is less than the theoretical amount that you could get. Percent Yield The ratio of the actual yield to the theoretical yield, multiplied by 100. actual % yield = theoretical x 100 Sample Problem 9-8 Chlorobenzene is prepared by the following process: C6H6 (l) + Cl2 (g) → C6H5Cl (s) + HCl (g) When 36.8 g of C6H6 react with an excess of Cl2, the actual yield of C6H5Cl is 38.8 g. What is the percent yield of chlorobenzene? Practice Problem #1 Methanol can be produced through the reaction of carbon monoxide and hydrogen in the presence of a catalyst. Pt CO (g) + 2 H2 (g) → CH3OH (l) If 75.0 g of CO reacts to produce 68.4 g of CH3OH, what is the percent yield of CH3OH? Practice Problem #2 Aluminum reacts with excess copper sulfate in the following unbalanced equation: Al (s) + CuSO4 (aq) → Al2(SO4)3 (aq) + Cu (s) If 1.85 g of Al react and the percent yield of Cu is 56.6 %, what mass of Cu is produced? Assignment – Due Monday BOP 9-3 Textbook Problems 9-3 Worksheet 9 Element Handbook Problems