biochem 41[3-12
advertisement

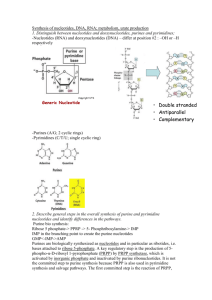
Chapter 41 Purine and Pyrimidine Metabolism Purines 1. What does alkaline phosphatase do in the intestines? dephosphorylates nucleotides to nucleosides 2. What is the first commited step of purine synthesis? Enzyme? What is added afterwards? What is the product after all these reactions? PRPP + glutamine’s amino group [glutamine is the mean glue that holds it together] i. catalyzed by glutamine phosphoribosyl amidotransferase ii. PRPP = 5-PhosphoRibosyl-1-PyroPhosphate Glycine, N10-formyl-FH4,glutamine, CO2, Aspartate, and another N10-formyl-FH4 i. 6 molecules of ATP are needed Inosine monophosphate (IMP) 3. 4. How is AMP made from IMP? How is GMP different (2 steps)? Aspartate + GTP is added by adenylosuccinate synthase, then fumarate is subtracted by adenylosuccinase IMP dehydrogenase turns IMP into xanthosine monophosphate; then Glutamine + ATP is added by GMP synthetase What four enzymes are regulated? How are they regulated? First 2 steps of IMP synthesis: PRPP synthetase, glutamine phosphoribosyl amidotransferase i. PRPP synthetase inhibited by ADP and/or GDP (2 allosteric sites) ii. The other enzyme is dimerized and inactivated by GMP and AMP First step of either AMP and GMP synth: Adenlyosuccinate synthetase, IMP dehydrogenase i. inhibited by AMP or GMP, respectively 5. SALVAGE. Occurs after 5’ nucleotidase digests DNA into nucleosides. What does purine nucleoside phosphorylase do? What is the second stage—how are nucleotides IMP or GMP reformed? What is special about the Adenine/AMP/Adenosine pathway? Replaces a purine base for a phosphate group, creating a free base: i. Guanosine is converted to guanine ii. Inosine to hypoxanthine (adenosine is first deaminated to inosine) Then, a phosphoribosyl transferase (HGPRT) takes phosphate from PRPP to reform the nucleotide, IMP or GMP i. The reactions in #3 convert IMP to AMP Adenosine can also be directly converted back to AMP by adenosine kinase 6. What happens when purine nucleoside phosphorylase is deficient due to a mutation? What do you call the condition caused by defective HGPRT? Clinical presentation? T cell immunity is compromised Lesch-Nyhan syndrome: mental retardation and self-mutilation Speculation: since de novo synthesis occurs in liver, brain, and leukocytes…maybe overactivation of this pathway is harming these tissues? Pyrimidines 7. What molecules are combined to make pyrimidines? What enzyme catalyzes the synthesis of Glutamine, CO2, and ATP into carbamoyl phosphate? What activates this reaction and where does it take place? Aspartate and carbamoyl phosphate CPSII: Carbamoyl phosphate synthetase II PRPP; Cytosol i. a MAP kinase makes CPSII more sensitive to PRPP approaching S phase 8. 9. What is the last mainline product, and what provides feedback inhibition to CPSII? UDP UTP How is CTP made? UTP + glutamine’s amino group [the glue of nucleotide synthesis] Does not work with anything but UTP 10. What does pyrimidine nucleoside phosphorylase do? What is the next step? Non-specific enzyme that converts pyrimidine bases into their respective nucleosides i. NOTE: purine version runs in opposite direction Nucleoside kinases make nucleosides into nucleotides Much simpler salvage than w/purines! Base=> nucleoside=> nucleotide 11. What is hereditary orotic aciduria, what causes it, and how is it cured? [test Q] Lack of enzymes converting orotate to UMP (defective orotate phosphoribosyltransferase and orotidine 5’ phophate decarboxylase) pyrimidines can’t be synthesized—synthesis is stuck at the intermediate orotate Administer uridine, which can be converted to UMP by other enzymes 12. What is another way that orotic aciduria can occur? [test Q] OTC (urea cycle) deficiency leaves too much carbamoyl phosphate floating around; this is used in pyrimidine production, bypassing the rate controlled step; lots of orotate is produced along this route, it just can’t be converted quickly enough to nucleotides 13. What does ribonucleotide reductase do? How does an accumulation of dTTP change its activity? How does an accumulation of dGTP change its activity? converts the ribose of a nucleotide (diphosphate nucleotides only!) into deoxyribose favors reduction of GDP to dGDP Teegax Favors reduction of ADP to dADP cycle of favor: T=> G=> A=> C or U 14. What can generally stimulate purine synthesis, and thereby cause gout? PRPP accumulation (used in first commited step), perhaps caused by lack of the enzyme glucose 6-phosphatase 15. What can be radiolabeled to determine if purines are being overproduced? 15N-labeled glycine 16. What does allopurinol inhibit? Suicide inhibitor of xanthine oxidase Also decreases synthesis by decreasing levels of PRPP (salvage of xanthine requires PRPP) 17. What happens when adenosine deaminase is deficienct? SCID [path: this is actually the less common variant of SCID]