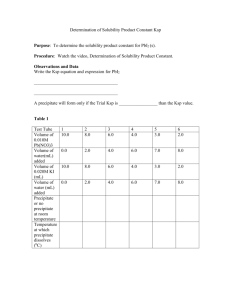
Chem 265 Quiz: Solubility, pH, and Equilibrium
advertisement

Wednesday Quiz 5 [3] 1. Chem 265 2012 What concentration of oxalate is required to precipitate 99.0% of 0.01 M Ce3+ ? Ksp for Ce2(C2O4)3 = 5.9 x 10-30 Amount remaining = 1.0 % 0.01 *0.01 M Ce3+ remains For this concentration, [C2O4]3 = [C2O4] = 8 x 10-8 M 1 sig fig [1] 2. Which is the stronger base of the following two? Urea Kb = 1.5 x 10-14 Hydrazine Kb = 1.1 x 10-6 5.9 𝑥 10−30 0.00012 =5.9 x 10-22 M [2] 3. If a 0.100 M solution of NaOH is added to a solution containing 0.200 M Ni 2+, 0.200 M Ce3+, and 0.200 M Cu2+, which metal hydroxide will precipitate first? Ksp for Ni(OH)2 = 6.0 X 10-16, Ksp for Ce(OH)3 = 6.0 X 10-22, and Ksp for Cu(OH)2 = 4.8 x 10-20. (a) Ni(OH)2 (b) Ce(OH)3 (c) Cu(OH)2 You need to calculate the solubility of each to decide which will ppt first [1] 4. What is the pH of a 0.100 molar solution of sodium chloroacetate. Chloroacetic acid is a moderately weak acid (a) 1.17 x 10-8 (b) 7.93 (c) 1.93 The salt of a weak acid is basic [3] 4. Find the concentration of Ag+ in equilibrium with AgBr(s) and 0.20 M Br-. Ksp for AgBr = 5.0 x 10-13 Ksp for AgBr = 5.0 x 10-13 = [Ag+] *[Br-] Let s = solubility = [Ag+] [Br-] = s +0.20 M ~ 0.20 M [Ag+] = 5x10−13 0.20 = 25 x 10-13 = 2.5 x 10-12 M