NOTES on AMMONIA
advertisement

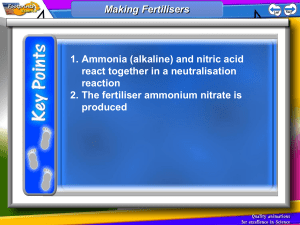
Ammonia Are produced from nitrogen reacted with hydrogen (Haber process). Used for producing: fertilisers, nitric acid, nylon, dyes, cleaners and dry cell. Objectives: Describe the use of nitrogen, from air, and hydrogen, from cracking oil, in the manufacture of ammonia. Describe and explain the essential conditions for the manufacture of ammonia by the Haber process. The Manufacture of Ammonia: The Haber Process The Process Nitrogen and hydrogen are mixed together in ratio 1:3, where nitrogen is obtained from fractional distillation of liquid air and hydrogen is obtained from natural gas (methane) with steam, or from cracking oil fractions, and passed over iron catalyst. Properties and Uses of Ammonia Physical properties: Colourless gas Characteristic pungent smell Non-poisonous, but can have adverse effects on the respiratory system if inhaled in large quantities Lighter than air Easily liquefiable even at low temperatures Boiling point –33.5° C Freezing point – 77.7° C Highly soluble in water Causes irritating burning sensation in nasal passage. Haber process Equation Since the reaction is reversible so H2 and N2, reproduced from decomposition of produced NH3, are passed over the catalyst again to produce ammonia. Ammonia as Fertilizers Objectives: Ammonia and its Uses Page 1 of 2 Describe the use of nitrogenous fertilisers in promoting plant growth and crop yield. Compare nitrogen content of salts used for fertilisers by calculating percentage masses. Describe eutrophication and water pollution problems caused by nitrates leaching from farm land and explain why the high solubility of nitrates increases these problems. Describe the displacement of ammonia from its salts and explain why adding calcium hydroxide to soil can cause the loss of nitrogen from added nitrogenous fertiliser. Chemistry Dept/Grade9/First Term/2014 Imaduddin School Plants need nitrogen as one of component for growth and ammonium fertilizers contain Nitrogen for that. groundwater is our drink source, when humans drink this water, they will get seriously ill and babies may suffer breathlessness to death. The Effect of Calcium Hydroxide Ammonium Fertilizer on Soil Soil that is too acidic presents different problems for crops. Calcium hydroxide, which is produced by adding water to calcium oxide, neutralises the acidity of the soil by reacting but it also react with ammonium fertilizers. So, Nitrogen is lost as ammonia gas escape fro m the soil. 2NH4NO3+ Ca(OH)2 --> 2NH3(g) + Ca(NO3)3 + 2H2O Chemical properties of Ammonia Problems with Ammonia Eutrophication is the inrease in organic content of water when fertilizers leach into soil and washed into rivers and streams. When excess fertilizers washed away by rain, nitrate ions in it gets into rivers and helps aquatic plants like algae to grow swiftly. When too much algae, water turns murky and sunlight would not penetrate into water to help their growth which in turn lead to deaths of algae. Decay of these algae uses up oxygen, hence killing aquatic animals. Then even more algae dies and even more animals die. Gives white fumes with hydrochloric acid Alkaline gas (turns red litmus paper blue) Neither combustible nor a supporter of combustion Highly soluble in water Forms salts with acids Reduces metal oxides to free metals Reacts with carbon dioxide to form urea Forms a pale blue copper hydroxide with copper sulphate Reduces halogens to halide ions Forms complex compounds with aqueous solutions of salts of copper or silver Water pollution results from runoff of fertilizer use, leaching from septic tanks, sewage and erosion of natural deposits. Nitrate ions from nitrogen in soil leaches down the soil into groundwater due to its solubility. Since Page 2 of 2 Chemistry Dept/Grade9/First Term/2014 Imaduddin School