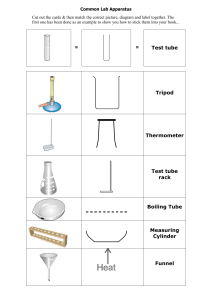
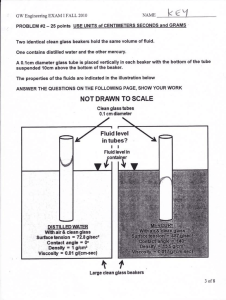
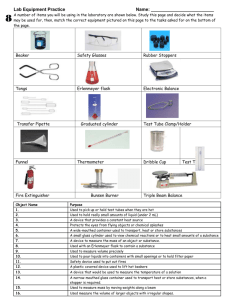
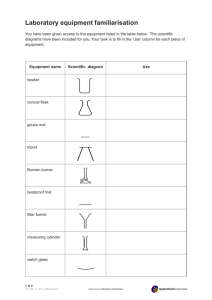
How to Apply Safety Techniques in the Lab Assignment
advertisement

How to Apply Safety Rules and Techniques for the Chemistry Laboratory This exercise explores some basic safety and first aid rules and the reason for their existence. In addition, you will also devise some techniques for doing some experiments. Use complete sentences to answer the questions below. PART A 1. Describe some clothing and accessories that would be inappropriate in the lab. Explain why they should NOT be worn. 2. Are there any other things associated with you person that might be a safety hazard in the lab? 3. Explain the location and the use of each with respect to the chem. lab: a. Fire extinquisher b. Fire blanket c. Phone d. Eyewash e. Shower 4. Explain what should be done if someone happens to: a. Receive a burn b. Receive a cut c. Pour acid or base on their body or clothing d. Cause a fire in the lab e. Get a chemical in his/her eyes f. Spill a large quantity of chemical liquid in the lab PART B Devise a procedure for the following situations. In each case identify all equipment necessary to carry out the procedure. 1. Test the odor of something 2. Pour a liquid from one beaker to another 3. Prepare a crucible and cover for lab use 4. Set up a hot water bath- with burner and with a hot plate 5. Set up an ice bath 6. Set up a filtration system- gravity and vacuum 7. Light a laboratory burner and obtain the best flame for lab work 8. Decant a liquid 9. Properly use a wash bottle 10. Cut glass 11. Bend glass 12. Fire polish glass 13. Use a transfer pipet 14. Read a buret 15. Read a graduated cylinder 16. Record measurement of mass 17. Heat a test tube attached to a ring stand 18. Heat a test tube using a test tube holder 19. Dispose of water-both liquid and solid 20. Determine the volume of a solid and of a liquid 21. Determine the density of a solid and of a liquid 22. Use a laboratory thermometer- regular and CBL 23. Empty hot liquid from a beaker 24. Evaporate water from a solution in a quantitative experiment 25. Determine the solubility of a substance 26. Collect gas by displacement of water 27. Titrate an acid with a base 28. Make 250 mL of a 0.5 M NaCl solution 29. Prepare a graph of the pH of an acid solution as you add base in 0.5 mL increments. 30. Record any laboratory measurement.