Chapter 7 2_11_15
advertisement

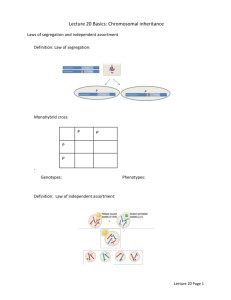
Chapter 7. Indirect impacts of coevolution ARGUE THAT THE STRENGTH OF SELECTION IS KEY. THIS COMPLEMENTS ARGUMENTS IN THE PRECEEDING CHAPTER THAT GENETIC ARECHITECTURE IS CRITICAL. LEADS TO A FINAL SYNTHESIS SUGGESTING THAT WE NEED TO KNOW TWO THINGS: 1) INFECTION GENETICS and 2) STRENGTH OF COEVOLUTION. WE KNOW NEITHER. FROM QUALITATIVE QUANTITATIVE AND PREDICTIVE Biological Motivation A long-standing mystery in evolutionary biology is the origin and maintenance of sexual reproduction (REFS). One of the key reasons sexual reproduction is so perplexing is that it should generally impose a twofold cost associated with the production of male offspring (REFS). One of the key benefits culd be recombination… But even that is a bit perplexing because recombination breaks apart co-adapted gene complexes. Thus, it would seem the benefits of recombination could accrue only in a changing environment. Arguments like these led Jaenike and Hamilton to propose the Red Queen hypothesis which suggested much of the reason for sex may be evading parasites. One of the best studied systems focused on elucidating the role of coevolution in sex is the interaction between snails and trematodes. These snails live within lakes distributed throughout New Zealand andWhat makes this interaction so useful is the presebce of sexual and asexual populations and variation in levels of trematode infection. Key Questions: Does coevolution between snail and trematode favor increased host recombination? What impact does coevolution have on rates of recombination in the parasite? Building a model of coevolution between snails and trematodes In order to understand how recombination rates evolve in response to coevolution, we must find a way to integrate genetic variation for recombination rates into the two locus models we studied in the previous chapter. The simplest way to accomplish this goal is to integrate a third locus, M, that modifies the rate of recombination between the coevolving loci A and B (Figure 1). Even if we limit ourselves to studying only haploid diallelic loci, we must now confront the challenges of studying a coevolutionary model with eight possible genotypes in each species. Because the equations get burly and long and complex, we are going to assume a particular infection matrix right off the bat. This costs us generality, but it will be paid for with clarity. The particular assumption we are going to make to adapt this interaction to the two-locus modeling framework we developed earlier in this chapter is that each genotype makes a FREP or mucin molecule with a unique Mathematica Resources: http://www.webpages.uidaho.edu/~snuismer/Nuismer_Lab/the_theory_of_coevolution.htm conformation. JUSTIFY USING MOLECULAR SPECIFICS… I THINK THIS MEANS YOU MUST HAVE AN ACTIVE SITE THAT IS ENCODED BY TWO SEPARATE LOCI? If the genotypes of snail and shistosome match, the host FREP molecule binds to the parasite mucin molecule and the infection is cleared. If, in contrast, the genotypes of the two species do not match, the snail FREP fails to bind to the schistosome mucin and the infection succeeds. Together, these assumptions lead to the following interaction matrix describing the probability that a schistosome genotype evades recognition and successfully infects a host genotype: 0 1 𝛼=[ 1 1 1 0 1 1 1 1 0 1 1 1 ] 1 0 We imagine that snails and trematodes run into each other at random, and that when a snail individual with genotype i encounters a trematode individual with genotype j, an infection results with probability 𝛼𝑖,𝑗 . If we assume that infection reduces the fitness of snails by some amount sx, the fitness of the eight possible snail genotypes is given by: 𝑊𝑋,𝐴𝐵● = 1 − 𝑠𝑋 ((𝑌𝐴𝑏𝑀 + 𝑌𝐴𝑏𝑚 ) + (𝑌𝑎𝐵𝑀 + 𝑌𝑎𝐵𝑚 ) + (𝑌𝑎𝑏𝑀 + 𝑌𝑎𝑏𝑚 )) (1a) 𝑊𝑋,𝐴𝑏● = 1 − 𝑠𝑋 ((𝑌𝐴𝐵𝑀 + 𝑌𝐴𝐵𝑚 ) + (𝑌𝑎𝐵𝑀 + 𝑌𝑎𝐵𝑚 ) + (𝑌𝑎𝑏𝑀 + 𝑌𝑎𝑏𝑚 )) (1c) 𝑊𝑋,𝑎𝐵● = 1 − 𝑠𝑋 ((𝑌𝐴𝐵𝑀 + 𝑌𝐴𝐵𝑚 ) + (𝑌𝐴𝑏𝑀 + 𝑌𝐴𝑏𝑚 ) + (𝑌𝑎𝑏𝑀 + 𝑌𝑎𝑏𝑚 )) (1e) 𝑊𝑋,𝑎𝑏● = 1 − 𝑠𝑋 ((𝑌𝐴𝐵𝑀 + 𝑌𝐴𝐵𝑚 ) + (𝑌𝐴𝑏𝑀 + 𝑌𝐴𝑏𝑚 ) + (𝑌𝑎𝐵𝑀 + 𝑌𝑎𝐵𝑚 )) (1g) where the ● indicates either the M or m allele at the modifier locus which has no impact on fitness. Similarly, if we assume encountering a resistant snail reduces the fitness of trematode by some amount sy, the fitness of the eight possible trematode genotypes is given by: 𝑊𝑌,𝐴𝐵● = 1 − 𝑠𝑌 (𝑋𝐴𝐵𝑀 + 𝑋𝐴𝐵𝑚 ) (2a) 𝑊𝑌,𝐴𝑏● = 1 − 𝑠𝑌 (𝑋𝐴𝑏𝑀 + 𝑋𝐴𝑏𝑚 ) (2c) 𝑊𝑌,𝑎𝐵● = 1 − 𝑠𝑌 (𝑋𝑎𝐵𝑀 + 𝑋𝑎𝐵𝑚 ) (2e) 𝑊𝑌,𝑎𝑏● = 1 − 𝑠𝑌 (𝑋𝑎𝑏𝑀 + 𝑋𝑎𝑏𝑚 ) (2g) Now, if we assume that the probability of survival to mating for the various snail and trematode genotypes depends on these fitnesses, we can calculate the frequency of each genotype after selection but prior to random mating. As before, we can calculate these frequencies by multiplying the current frequency by its relative fitness. This yields the following expressions for snail and trematode genotype frequencies after selection: 2 𝑋𝑖′ = 𝑋𝑖 𝑊𝑋,𝑖 ̅𝑋 𝑊 (3a) 𝑌𝑖′ = 𝑌𝑖 𝑊𝑌,𝑖 ̅𝑌 𝑊 (3b) ̅𝑋 and 𝑊 ̅𝑌 , are given by: where the population mean fitnesses, 𝑊 ̅𝑋 = ∑8𝑖=1 𝑋𝑖 𝑊𝑋,𝑖 𝑊 (4a) ̅𝑌 = ∑8𝑖=1 𝑌𝑖 𝑊𝑌,𝑖 𝑊 (4b) We now know the frequency of each of the eight genotypes within both host and parasite after species interactions have occurred. Next, we must predict how these genotype frequencies will change in response to a single round of random mating and recombination. Conceptually, integrating random mating and recombination is a simple matter. We need only sum up the frequency with which all possible matings occur, and weight each possible mating the by the probability that it produces a specific offspring genotype. Mathematically, this reasoning leads to the following expressions for the frequency of genotype i after random mating and recombination in snail and trematode: 𝑋𝑖′′ = ∑8𝑗=1 ∑8𝑘=1 𝑋𝑗′ 𝑋𝑘′ Π𝑋,𝑗+𝑘→𝑖 (5a) 𝑌𝑖′′ = ∑8𝑗=1 ∑8𝑘=1 𝑌𝑗′ 𝑌𝑘′ Π𝑌,𝑗+𝑘→𝑖 (5b) where the terms Π𝑋,𝑗+𝑘→𝑖 and Π𝑌,𝑗+𝑘→𝑖 are the probabilities that parental genotypes j and k produce offspring genotype i in snail and trematode, respectively. Although these equations appear quite simple, they actually mask a huge amount of biological detail and tedious calculation. Specifically, there are 64 different Π𝑋,𝑗+𝑘→𝑖 terms and 64 different Π𝑌,𝑗+𝑘→𝑖 terms that must be worked out, each of which is a function of the recombination rates between the A and B loci and the B and M loci. In the previous chapter, we tackled this problem by creating a table depicting the frequency of offspring produced by each possible mating. Although this was quite manageable with only a pair of loci, it becomes very unwieldy for three loci because there are 64 possible types of matings, each of which can produce 8 possible offspring genotypes. As much fun as it would be to develop such a table, what would be the point when it wouldn’t even fit on a single page? Instead, it is much more practical (and accurate) to develop a simple computer algorithm that generates this table automatically. In the accompanying Mathematica notebook, just such an algorithm is developed and used to perform the calculations proscribed by equations (5). At this point, we have a really huge algebraic mess on our hands. It is no understatement to say that writing out the fully expanded versions of equations (5) would yield expressions of such length and tedium that we would almost certainly cry blood. So what can we do? Perhaps the single most powerful 3 approach we can employ at this point to make sense out of these complex expressions is the QuasiLinkage Equilibrium (QLE) approximation we introduced in the previous chapter. Analyzing the model Although the general assumptions of our QLE approximation mirror those of the previous chapter, the specific steps we must take to implement the approximation are now a bit more complex. One significant change from the previous chapter is that in order to change variables from genotype frequencies to statistical moments, we now need more than a pair of allele frequencies and a single measure of linkage disequilibrium to fully describe the system. For each species, we now need to follow three allele frequencies, three pairwise linkage disequilibria, and one three-way linkage disequilibrium. The recursions for these new variables are: ′′ ′′ ′′ ′′ ′′ 𝑝𝑋,𝐴 = 𝑋𝐴𝐵𝑀 + 𝑋𝐴𝐵𝑚 + 𝑋𝐴𝑏𝑀 +𝑋𝐴𝑏𝑚 (6a) ′′ ′′ ′′ ′′ ′′ 𝑝𝑋,𝐵 = 𝑋𝐴𝐵𝑀 + 𝑋𝐴𝐵𝑚 + 𝑋𝑎𝐵𝑀 +𝑋𝑎𝑏𝑚 (6b) ′′ ′′ ′′ ′′ ′′ 𝑝𝑋,𝑀 = 𝑋𝐴𝐵𝑀 + 𝑋𝐴𝑏𝑀 + 𝑋𝑎𝐵𝑀 +𝑋𝑎𝑏𝑀 (6c) ′′ ′′ ′′ ) ′′ ′′ 𝐷𝑋,𝐴𝐵 = (𝑋𝐴𝐵𝑀 + 𝑋𝐴𝐵𝑚 − 𝑝𝑋,𝐴 𝑝𝑋,𝐵 (6d) ′′ ′′ ′′ ) ′′ ′′ 𝐷𝑋,𝐵𝑀 = (𝑋𝐴𝐵𝑀 + 𝑋𝑎𝐵𝑀 − 𝑝𝑋,𝐵 𝑝𝑋,𝑀 (6e) ′′ ) ′′ ′′ ′′ ′′ 𝐷𝑋,𝐴𝑀 = (𝑋𝐴𝐵𝑀 + 𝑋𝐴𝑏𝑀 − 𝑝𝑋,𝐴 𝑝𝑋,𝑀 (6f) ′′ ′′ ′′ ′′ ′′ ′′ ′′ ′′ ′′ ′′ 𝐷𝑋,𝐴𝐵𝑀 = 𝑋𝐴𝐵𝑀 − 𝐷𝑋,𝐴𝐵 𝑝𝑋,𝑀 − 𝐷𝑋,𝐴𝑀 𝑝𝑋,𝐵 − 𝐷𝑋,𝐵𝑀 𝑝𝑋,𝐴 − 𝑝𝑋,𝐴 𝑝𝑋,𝐵 𝑝𝑋,𝑀 (6g) where blah blah blah. The next step, in our change of variables is to substitute all expressions involving the old variables X on the right hand sides of these expressions with the definitions of these old variables in terms of the new variables. To do this, we can use the following formulae: 𝑋𝐴𝐵𝑀 = 𝑝𝑋,𝐴 𝑝𝑋,𝐵 𝑝𝑋,𝑀 + 𝐷𝑋,𝐴𝐵 𝑝𝑋,𝑀 + 𝐷𝑋,𝐴𝑀 𝑝𝑋,𝐵 + 𝐷𝑋,𝐵𝑀 𝑝𝑋,𝐴 + 𝐷𝑋,𝐴𝐵𝑀 𝑋𝐴𝐵𝑚 = 𝑝𝑋,𝐴 𝑝𝑋,𝐵 𝑞𝑋,𝑀 + 𝐷𝑋,𝐴𝐵 𝑞𝑋,𝑀 − 𝐷𝑋,𝐴𝑀 𝑝𝑋,𝐵 − 𝐷𝑋,𝐵𝑀 𝑝𝑋,𝐴 − 𝐷𝑋,𝐴𝐵𝑀 𝑋𝐴𝑏𝑀 = 𝑝𝑋,𝐴 𝑞𝑋,𝐵 𝑝𝑋,𝑀 − 𝐷𝑋,𝐴𝐵 𝑝𝑋,𝑀 + 𝐷𝑋,𝐴𝑀 𝑞𝑋,𝐵 − 𝐷𝑋,𝐵𝑀 𝑝𝑋,𝐴 − 𝐷𝑋,𝐴𝐵𝑀 𝑋𝐴𝑏𝑚 = 𝑝𝑋,𝐴 𝑞𝑋,𝐵 𝑞𝑋,𝑀 − 𝐷𝑋,𝐴𝐵 𝑞𝑋,𝑀 − 𝐷𝑋,𝐴𝑀 𝑞𝑋,𝐵 + 𝐷𝑋,𝐵𝑀 𝑝𝑋,𝐴 + 𝐷𝑋,𝐴𝐵𝑀 𝑋𝑎𝐵𝑀 = 𝑞𝑋,𝐴 𝑝𝑋,𝐵 𝑝𝑋,𝑀 − 𝐷𝑋,𝐴𝐵 𝑝𝑋,𝑀 − 𝐷𝑋,𝐴𝑀 𝑝𝑋,𝐵 + 𝐷𝑋,𝐵𝑀 𝑞𝑋,𝐴 − 𝐷𝑋,𝐴𝐵𝑀 𝑋𝑎𝐵𝑚 = 𝑞𝑋,𝐴 𝑝𝑋,𝐵 𝑞𝑋,𝑀 − 𝐷𝑋,𝐴𝐵 𝑞𝑋,𝑀 + 𝐷𝑋,𝐴𝑀 𝑝𝑋,𝐵 − 𝐷𝑋,𝐵𝑀 𝑞𝑋,𝐴 + 𝐷𝑋,𝐴𝐵𝑀 𝑋𝑎𝑏𝑀 = 𝑞𝑋,𝐴 𝑞𝑋,𝐵 𝑝𝑋,𝑀 + 𝐷𝑋,𝐴𝐵 𝑝𝑋,𝑀 − 𝐷𝑋,𝐴𝑀 𝑞𝑋,𝐵 − 𝐷𝑋,𝐵𝑀 𝑞𝑋,𝐴 + 𝐷𝑋,𝐴𝐵𝑀 𝑋𝑎𝑏𝑚 = 𝑞𝑋,𝐴 𝑞𝑋,𝐵 𝑞𝑋,𝑀 + 𝐷𝑋,𝐴𝐵 𝑞𝑋,𝑀 + 𝐷𝑋,𝐴𝑀 𝑞𝑋,𝐵 + 𝐷𝑋,𝐵𝑀 𝑞𝑋,𝐴 − 𝐷𝑋,𝐴𝐵𝑀 Identical formulae hold for the parasite as well… 4 Finally, we QLE the shit out of it. ALTHOUGH I HAVE WRITTEN ALL OF THESE OUT, IT IS FAR EASIER TO UNDERSTAND THEM IN A GENERAL SENSE BY CONSULTING THE WONDERFUL PAPER BY KJB WHICH PROVIDES GENERAL FORMULAE. Although this change of variables should be mostly familiar after the previous chapter, we do encounter a novel association in the three way. What does this mean? OK, NOW WHAT WE HAVE IS A NICE TIDY SYSTEM OF EQUATIONS DESCRIBING HOW ALLELE FREQUENCIES AND DISEQUILIBRIA CHANGE IN EACH OF OUR INTERACTING SPECIES. UNFORTUNATELY, there is still a catch: these equations remain ten miles long. So, let’s now apply our QLE approximation which should hold as long as recombination is relatively frequent relative to the strength of selection (REF). IF WE DO WHAT WE DID IN THE LAST CHAPTER, Mechanistically, this means that we Taylor Expand our expressions for evolutionary change ignoring all terms that are of order eps or greater which in this case includes terms like s^2, D^2, and s*D and higher. IF WE DO THAT NOW WE FIND THAT: ∆𝑝𝑋,𝑀 ≈ 0 ∆𝑝𝑌,𝑀 ≈ 0 WHY DOES OUR MODIFIER NOT CHANGE??? BLAH BLAH BLAH if we go out to the next order we see that… ∆𝑝𝑋,𝑀 ≈ 𝑎𝑋,𝐴 𝐷𝑋,𝐴𝑀 + 𝑎𝑋,𝐵 𝐷𝑋,𝐵𝑀 + 𝑎𝑋,𝐴𝐵 𝐷𝑋,𝐴𝐵𝑀 where 𝑎𝐴 measures the strength of directional selection acting on locus A and is given by: 𝑎𝑋,𝐴 = −𝑠𝑋 (1 − 2𝑝𝑌,𝐴 )(1 − 𝑝𝑌,𝐵 − 𝑝𝑋,𝐵 (1 − 2𝑝𝑌,𝐵 )) 𝑎𝐵 measures the strength of directional selection acting on locus B and is given by: 𝑎𝑋,𝐵 = −𝑠𝑋 (1 − 2𝑝𝑌,𝐵 )(1 − 𝑝𝑌,𝐴 − 𝑝𝑋,𝐴 (1 − 2𝑝𝑌,𝐴 )) and 𝑎𝐴𝐵 measures the strength of epistatic selection acting on locus A and B together and is given by: 𝑎𝑋,𝐴𝐵 = 𝑠𝑋 (1 − 2𝑝𝑌,𝐴 )(1 − 2𝑝𝑌,𝐵 ) AND FOR SPECIES Y if we go out to the next order we see that… ∆𝑝𝑌,𝑀 ≈ 𝑎𝑌,𝐴 𝐷𝑌,𝐴𝑀 + 𝑎𝑌,𝐵 𝐷𝑌,𝐵𝑀 + 𝑎𝑌,𝐴𝐵 𝐷𝑌,𝐴𝐵𝑀 where 𝑎𝐴 measures the strength of directional selection acting on locus A and is given by: 5 𝑎𝑌,𝐴 = 𝑠𝑌 (1 − 2𝑝𝑋,𝐴 )(1 − 𝑝𝑌,𝐵 − 𝑝𝑋,𝐵 (1 − 2𝑝𝑌,𝐵 )) 𝑎𝐵 measures the strength of directional selection acting on locus B and is given by: 𝑎𝑌,𝐵 = 𝑠𝑌 (1 − 2𝑝𝑋,𝐵 )(1 − 𝑝𝑌,𝐴 − 𝑝𝑋,𝐴 (1 − 2𝑝𝑌,𝐴 )) and 𝑎𝐴𝐵 measures the strength of epistatic selection acting on locus A and B together and is given by: 𝑎𝑌,𝐴𝐵 = −𝑠𝑌 (1 − 2𝑝𝑋,𝐴 )(1 − 2𝑝𝑋,𝐵 ) SO WHAT CAN WE LEARN FROM THIS EXPRESSion? Explain the different forces acting on the modifier… POINT OUT HOW THIS EQUATION SHOWS THAT MODIFIER WOULD INCREASE IF SIGN OF LD AND E WAS OPSSITE. But what would these values be??? NOW INTRODUCE QLE RECURSIONS FOR MOMENTS AND SOLVE FOR THEIR QLE VALUES… 𝑀𝑚 ∆𝐷𝑋,𝐴𝑀 ≈ −(𝑟𝑋,𝐴𝐵 + 𝑟𝑋,𝐵𝑀 )𝐷𝑋,𝐴𝑀 ∆𝐷𝑋,𝐵𝑀 ≈ −(𝑟𝑋,𝐵𝑀 )𝐷𝑋,𝐵𝑀 ∆𝐷𝑋,𝐴𝐵 ≈ −(𝑟𝑋,𝐵𝑀 )𝐷𝑋,𝐴𝐵 WHAT A MESS… WHICH FORM SHOULD I WRITE THE r’s IN? AS DELTA OR NOT? SHOULD I IGNORE DOUBLE RECOMBO’s? I did above… Answers to key questions New Questions Arising: What if infection genetics differs? What happens when selection is strong and recombination weak? Extensions Extension 1: Alternative infection matrices Generalization 2: Strong selection and weak initial recombination 6 Conclusions and Synthesis For the Red Queen to favor more sex, selection must be strong. No matter what the form of genetic interaction, weak selection just won’t cut it. Interestingly, in the interaction between snails and trematodes, cycles look fast (insert figure). In fact, they look fast like strong selection fast. Thus, this system might be one where the red queen is in operation. However, there is little evidence to suggest coevolution is always so strong. Thus, it seems unlikely that the red queen is a general explanation for the evolution of sex. Conclusions and Synthesis 7 References Figure Legends Dybdahl, M. F., C. E. Jenkins, and S. L. Nuismer. 2014. Identifying the Molecular Basis of Host-Parasite Coevolution: Merging Models and Mechanisms. AMERICAN NATURALIST 184:1-13. Mitta, G., C. M. Adema, B. Gourbal, E. S. Loker, and A. Theron. 2012. Compatibility polymorphism in snail/schistosome interactions: From field to theory to molecular mechanisms. Developmental and Comparative Immunology 37:1-8. 8 9