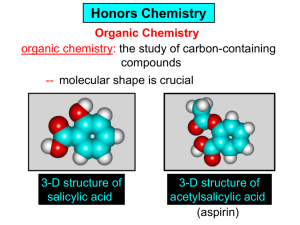
Organic Chemistry Unit Test SCH4U Spring 2012 Name: Date: May
advertisement

Organic Chemistry Unit Test SCH4U Spring 2012 Name: Date: May 30, 2012 K= /20 I= /26 MC = /9 C= /12 Part A: Multiple Choice (K = 20) Please put the letter that corresponds to the answer on the line before the question 1. ______ The compound above is classified as a(n) a) ketone b) carboxylic acid c) aldehyde d) ester 2. ______ Which of the following compounds is saturated? a) propene b) octane c) but-2-ene d) hept-3-yne 3. ______ Which formula represents an alkyne? a) C5H8 b) C5H12 d) C5H10 c) C5H16 4. ______ What is the name of the alkyl group containing four carbon atoms? a) propyl b) methyl c) butyl d) pentyl 5. ______ Which of the following is the correct name for the structure to the right? a) trans-4-nonyne b) cis-5-nonene c) trans-4-nonene d) cis-4-nonene 6. ______ Which of the following compounds is considered to be non-polar? a) CH3CH2OH b) CH3CHO c) CH3CH2NH2 d) CH3CH2CH3 7. ______ Which of the following compounds is a secondary alcohol? a) 1-pentanol b) 2-pentanone c) 2-pentanol d) 2-methyl-2-butanol 8. ______ Which of the following molecules below has a chiral isomer? a) b) c) d) 9. ______ The molecule CH3COCH3 is classified as a(n) a) ether b) ester c) ketone d) carboxylic acid 10. ______ Esters are formed by reacting the following two species a) alkane, alkene b) carboxylic acid, alcohol c) alcohol, water d) 2 alcohols 11. ______ Which statement is true of the carbon-carbon bonds in benzene a) they are identical to the carbon-carbon bonds in cyclohexane b) they are identical to the carbon-carbon bonds in cyclohexene c) they are half-way between a double and single bond d) they are easily broken in chemical reactions 12. ______ Identify the set of compounds that have the same functional group a) C2H6, C2H4, C2H2 b) C2H2, CH3COOH, CH3CONH2 c) CH3CH2CHO, H2CO, CH3CH2CH2CH2CHO d) CH3NH2, CH3CH2CONH2, CH3CH2CONHCH3 13. ______ Ethyl methanoate is used as an artificial rum flavouring. What type of compound is it? a) carboxylic acid b) ester c) ether d) ketone 14. ______ Which compound is an ether? a) CH3OH b) CH3COCH3 c) CH3COH d) CH3CH2OCH3 15. ______ The name 2-methyl-4-butanal is incorrect. What is the correct IUPAC name for this compound? a) 4-methyl-2-butanal b) 3-methylbutanal c) 2-methylbutanal d) 2-methyl-1-butanal 16. ______ Which of the following compounds is unable to form hydrogen bonds with itself? a) CH3CH2CH2CH2OH b) CH3CH2NH2 c) CH3COOH d) CH3CH2COOCH2CH3 17. ______ What is the IUPAC name for the compound C2H5CONH2? a) butanamide b) propan-2-amine c) propanamide d) ethylpropanamide 18. ______ Which is a characteristic feature of a substitution reaction? a) carbon atoms are bonded to the same number of atoms in the product as in the reactant b) carbon atoms are bonded to a smaller number of atoms in the product than in the reactant c) carbon atoms are bonded to a larger number of atoms in the product than in the reactant d) two compounds react to form one compound 19. ______ In an oxidation reaction a) a carbon will form fewer bonds to carbon b) a carbon will form fewer bonds with oxygen or more bonds to hydrogen c) a carbon will form more bonds with oxygen or fewer to hydrogen d) two small molecules combine to form a larger molecule 20. ______ Which reaction would be performed to convert an alkyne to alcohol? a) substitution b) elimination c) addition d) hydrolysis Part B: Draw and name the missing reagent, reactant or product AND name the type of reaction. (I = 20) 1. 2. 3. + 4. 5. + H2O propanoic acid + ethan-1-amine Part C: Short Answer (I = 6, MC = 9, C = 12) 1. The structure given below is Penicillin G, an antibiotic. Circle the functional groups in the molecule and list their corresponding names. (I = 3) 2. Draw the structural isomers of pentane. (I = 3) 3. During a school laboratory test, you are given pent-2-yne and asked to prepare pentan-2-one and pentan-3one from it. Develop a synthesis pathway to accomplish this task. (MC = 9) 4. Why does the hydrogenation of but-2-yne with 1.0 mole of hydrogen produce 2 isomers? Name the isomers produced. (C = 3) 5. Explain the difference in reaction with a halogen and each of the following compounds: alkane, alkene and benzene. Make sure you indicate the type of reaction and the conditions necessary for each. Draw the reaction for each. (C = 9)