Answer Key Answer Key (continued) Answer Key (continued
advertisement

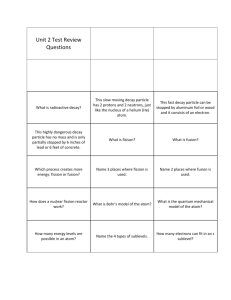
Answer Key Chapter 5 Practice Problems 1. 4.741014 Hz 2. 6.251010 m; (0.625 nm) 3. 3.05 m 4. 3.331013 J 5. Ephoton (violet) 4.51019 J Ephoton (red) 2.81019 J Edifference 1.71019 J 6. First calculate the frequency of the light. 6.121015 Hz Ephoton 4.061018 J 7. Bohr’s model showed that electrons move in circular orbits around the nucleus. The electrons have the energies associated with those orbits. 8. The electron is moving from an orbital of higher energy to an orbital of lower energy. 9. The second principal energy level can hold eight electrons (two in the 2s sublevel and six more in the 2p sublevel). The third principal energy level can hold eighteen electrons (two in the 3s sublevel, six in the 3p sublevel, and ten in the 3d sublevel). The difference is due to the fact that the larger third principal energy level has one more available energy sublevel. 10. The sublevels are written in order of increasing energy. 11. The number of electrons is indicated as a superscript integer to the right of the sublevel. 12. a. 1s22s22p63s23p4 c. 1s22s22p63s23p64s23d104p5 2 2 6 2 6 2 b. 1s 2s 2p 3s 3p 4s d. 1s22s22p63s2 Answer Key (continued) 13. a. b. c. d. e. 14. a. [He]2s22p5 [Ne]3s23p3 [Ar]4s2 [Ar]4s23d7 [Ar]4s23d104p4 c. f. g. h. i. j. e. [Kr]5s24d5 [Kr]5s24d105p5 [Xe]6s24f11 [Xe]6s24f145d7 [Rn]7s2 g. b. d. f. h. 15. eight dots surrounding the symbol for the element 16. Student responses may include any of the group 15 elements: nitrogen, phosphorus, arsenic, antimony, and bismuth. Chapter 5 Review 17. The equation c relates the speed, wavelength, and frequency of an electromagnetic wave. The frequency of a given wave cannot change; wave speed and wavelength are directly related. Therefore, the wavelength will decrease as speed decreases. 18. Light striking a metal plate dislodges electrons from the plate. Einstein concluded that light could act not only as a wave, but also as a stream of particles, called photons. 19. Energy increases as frequency increases. Energy decreases as wavelength increases (because frequency decreases). 20. According to Bohr’s model, an atom’s electrons travel around the nucleus in circular orbits, each of which has a specific energy value. Electrons can jump only from one allowable orbit to another. Therefore, electrons can increase or decrease in energy only by certain amounts. These amounts correspond to the frequencies (colors) of the light seen in the atomic emission spectrum. 21. Three energy sublevels are available—s, p, and d. An s sublevel can hold two electrons; a p sublevel can hold six electrons; and a d sublevel can hold ten electrons. Answer Key (continued) 22. In the modern model of the atom, electrons are not in specific orbits. Instead, they are in regions of space in which there is a high probability of finding electrons of a certain energy. 23. a. fluorine e. rhodium b. manganese f. polonium c. germanium g. sulfur d. sodium h. cadmium 24. The dots represent the electrons in the highest principal energy level of the atom. These electrons are called valence electrons, and they determine many of the chemical properties of an element. 25. Student responses may include helium and any of the group 2 elements: beryllium, magnesium, calcium, strontium, barium, and radium.