Answer Sheet
advertisement

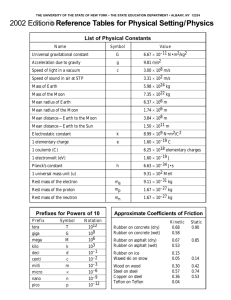
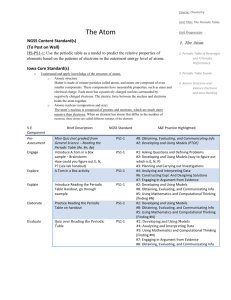
1. Put the following elements in order from smallest to largest atomic radius and explain why: C, O, Sn, Sr. O, C, Sn, Sr. Atomic radius increases as you move left and down the periodic table. 2. Put the following elements in order from lowest to highest first ionization energy and explain why: Al, Ar, Cs, Na. Cs, Na, Al, Ar. Ionization energy increases as you move up and to the right on the periodic table. 3. Which of the following elements most likely has the highest melting point – I, Mo, Te? Explain why. Mo. Melting point increases as you move towards the middle of the periodic table. 4. Which of the following elements is classified as a metal? – C, Xe, H, Na? Explain why. Na. Sodium is on the left side of the dark line on the periodic table. The only nonmetal on this side of the table is hydrogen. 5. Which of the following elements is classified as a nonmetal? – B, N, K, Fe? Explain why. N. Nitrogen is to the right of the dark line on the periodic table. Boron is on the line and is considered a metalloid. The other two choices are metals. 6. Which of the following elements is classified as a metalloid? – N, Si, Pb, Li? Explain why. Si. Silicon is located along the dark line that divides the periodic table. It has properties of both metals and nonmetals. 7. Which of the following elements is classified as a transition metal? – Ir, Bi, Mg, Ar? Explain why. Ir. It is located in the “transition” or bridge piece in the periodic table (The “B” groups) 8. Which of the following elements is classified as an inner transition metal? – Ir, Bi, Mg, U? Explain why U. It is located in one of the two rows that are pulled out of the periodic table and positioned on the bottom of the chart. These two rows contain many of the radioactive elements. 9. Which group contains the alkaline earth metals? Group 2A 10. Which group contains the alkali metals? Group 1A 11. Which group contains the chalcogens? Group 6A 12. Which group contains the halogens? Group 7A 13. Which group contains the noble gases? Group 8A 14. Which run left to right on the periodic table, periods or groups? Periods 15. Which can be used to determine the number of valence electrons in an atom, its period number or group number? Group number 16. Which can be used to determine the number of energy shells in an atom, its period number or group number? Period number Draw the orbital diagram and longhand for the following atoms: C Be 1s22s22p2 1s22s2 Ca 1s22s22p63s23p64s2 Li Si 1s22s 1s22s22p63s23p2 Draw the shorthand configuration for the following atoms: S Li K N F [Ne]3s23p4 [He]2s1 [Ar]4s1 [He]2s22p3 [He]2s22p5 Be able to choose the correct orbital configuration from a multiple choice question. Be able to draw and complete the bohr and Lewis dot diagram for an atom. Make sure you can pick out the correct one from a multiple choice question. c = λf E= hf c = 3.00 x 108 m s h = 6.6262 x 10 -34 J •s Hz = 1 s 17. A certain photon of light has a wavelength of 422 nm. What is the frequency of the light? λ = 422 nm (which is 0.000000422 m) f=? c = λf c=f λ (3.00 x 108m) = 7.11 x 1014 Hz (s)(0.000000422m) 18. What is the energy of the photon from question 17? -34 E= hf E = (6.6262 x 10 f = 7.11 x 1014 Hz or 7.11 x 1014 J • s)( 7.11 x 1014 ) s E = 4.71 x 10-19 J s 19. What is the energy of a quantum of light with a frequency of 7.39 X 1014 Hz? -34 E = (7.39 x 1014) (6.6262 x 10 E = hf J • s) (s) f = 7.39 X 1014 Hz or E = 4.90 x 10-17 J 7.39 x 1014 S 20. A certain blue light has a frequency of 6.91 X 1014 Hz. What is the wavelength of the light? c = λf rearranged to: f = 6.91 X 1014 Hz or (3.00 x 108m)(s) = λ (s)(6.91 x 1014) c=λ f = 6.91 X 1014 f s λ = 0.000000434m or 434 nm 21. The energy of a quantum of light is 2.84 X 10-19J. What is the frequency of the light? E = hf rearranged to: E= 2.84 X 10-19J (2.84 X 10-19J) =f (6.6262 x 10.34 J• s) E=f H f = 4.29 x 1014 s or 4.29 x 1014 Hz 22. Write the alpha decay equation for the following elements: U→ 238 4 92 2 He + Cm → 4 He + 243Pu 234 247 90 96 Th 2 94 23. Write the beta decay equation for the following elements: U→ 238 238 92 93 0 Np + Cm → e -1 247 247 Bk + 0 e 96 97 -1 24. Write the gamma decay equation for the following elements: U* → U+ 0ᵞ 238 238 92 92 0 Cm* → Cm + 0 ᵞ 247 247 96 96 0 25. What is the difference between fission and fusion? Fission splits a large, heavy nucleus (like Uranium) into two smaller, lighter nuclei. Fusion combines two small, light nuclei (like hydrogen), into a larger, more stable nucleus (like helium). 26. What are the three types of radiation/decay? Describe what happens to cause each type. Alpha radiation: Caused when the nucleus contains too many protons and the nuclear forces that hold them together “break”. A helium atom is expelled during the decay process. Beta radiation: Caused when the number of neutrons is too high compared to the number of protons. The atom converts a neutron into a proton and an electron. The proton stays in the atom, but the electron is expelled. Gamma radiation: Caused when an atom has too much energy and has become unstable. As it lowers back into a stable state of energy, photons of energy known as gamma rays are emitted. These are not particles that have mass, they are pure energy. 27. Be able to define wavelength, amplitude, frequency, crest, trough, origin (of a wave). Be able to pick out the correct diagrams if given multiple choice. Wavelength = the length of one complete wave Amplitude = the distance from the origin to the top of a crest or bottom of a trough Frequency = the number of waves that pass a certain point in a given amount of time. It is measure in Hz (hertz), or 1/s. Crest = a peak of a wave Trough = a valley of a wave Origin = the center line drawn directly through the middle of the wave. 28. Describe Planck and Einstein’s contributions to wave and particle science. Planck developed the quantum theory and planck’s constant. Einstein developed the Wave-Particle Duality theory, discovered the photoelectric effect, and developed the formula for deriving energy from the change in mass and speed of light (e = mc2). 29. Describe shielding and how it affects atomic radius. Shielding is when all the electrons inside the inner energy levels in an atom “block” the positive charge from the nucleus towards an electron in an outer energy level. As a result, that outer electron isn’t as attracted to the nucleus as the inner electrons, and it isn’t held as tightly. It can roam around a wider orbital or be more easily snatched off the atom by other atoms as a result of the weaker attraction to the positive nucleus. 30. Which have a larger radius, cations or their regular atoms? Their regular atoms. 30. Which have a larger radius, anions or their regular atoms? Anions