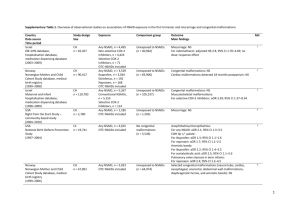
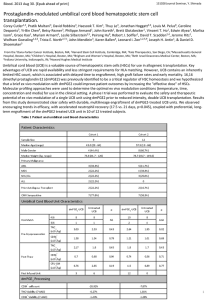
Supplementary Table 2. Overview of observational studies on associations of NSAID exposure in the second and/or third trimester and other pregnancy outcomes
Country
Study design
Exposure
Comparison
Outcome
Data source
Size
group
Main findings
Time period
USA
CC
Any NSAID, n = 33
No PPHN
PPHN: NS
Slone Epidemiology
n = 1,213
OTC NSAIDs included
(n = 836)
Center’s Birth Defects
Study
(1998–2003)
USA
CC
Any NSAID, n = 64
Unexposed to Quadriparetic CP in children born < 28 weeks after LMP:
The ELGAN Study
n = 877
OTC NSAIDs included
NSAIDs
For any NSAID: aOR 2.4; 95% CI 1.04–5.8
Children < 28 weeks old
(n = 813)
Diaparetic CP in children born < 28 weeks after LMP:
in 11 institutions
For any NSAID: aOR 3.5; 95% CI 1.1–11.0
(2002–2004)
USA
CH
Indomethacin, n = 85
Unexposed to Grade III or IV IVH in VLBW babies:
The NICHD Neonatal
n = 573
No OTC NSAIDs
indomethacin For indomethacin: aOR 2.7; 95% CI 1.2–6.4
Research Network’s Data
(n = 464)
Base Study
(1995–2000)
USA
CC
Any NSAID, n = 50
No PPHN
PPHN:
Hospital records,
n = 101
Ibuprofen, n = 23
(n = 61)
For any NSAID: aOR 21.5; 95% CI 7.1–64.7
meconium analysis
Naproxen, n = 19
For ibuprofen: aOR 12.9; 95% CI 3.9–42.3
(2001)
Acetylsalicylic acid, n =
For naproxen: aOR 3.3; 95% CI 1.2–9.3
44
For acetylsalicylic acid: aOR 8.1; 95% CI 3.3–20.0
OTC NSAIDs included
Finland
CC
Indomethacin, n = 31
No renal
Renal damage: NS
Record analysis
n = 66
No OTC NSAIDs
damage at
(2001)
< 33
gestational
weeks
(n = 35)
Finland
CH
Indomethacin, n = 82
None
Grade I or II IVH:
Record analysis
n = 240
No OTC NSAIDs
For indomethacin dose > 150mg/day: aOR 3.9; 95% CI 1.4–10.7
(2001)
For indomethacin therapy > 2 days: aOR 3.4; 95% CI 1.3–8.9
Severe NEC:
For indomethacin dose > 150mg/day: aOR 4.3; 95% CI 1.2–14.4, for
indomethacin therapy > 2 days: aOR 4.4; 95% CI 1.1–17.4
Sepsis:
For indomethacin: aOR 9.3; 95% CI 2.3–36.9
Ref.
1
2
3
4
5
6
USA
Record analysis
(1991–1995)
USA
Record analysis
(1986– 1991)
CH
n = 72
Indomethacin, n = 72
No OTC NSAIDs
None
Premature closure of ductus arteriosus:
For indomethacin with advancing gestational age, p < 0.05.
7
CC
n = 114
Indomethacin, n = 57
No OTC NSAIDs
Unexposed to
indomethacin
at
corresponding
gestational
age
(n = 57)
Increased risk of premature closure of ductus arteriosus:
For indomethacin: p < 0.05
NEC:
For indomethacin: p < 0.005
Grade II or IV IVH:
For indomethacin: p < 0.02
Oliguria:
For indomethacin: p < 0.003
8
Abbreviations: CH, cohort; CC, case–control; PPHN, persistent pulmonary hypertension of the newborn; NS, no excess risk (statistically not significant); ELGAN, Extremely Low Gestational Age Newborns;
CP, cerebral palsy; LMP, last menstrual period; aOR = adjusted OR; IVH = intraventricular haemorrhage; VLBW = very low birth weight (< 1500g); NEC = necrotizing enterocolitis.
1.
2.
3.
4.
5.
6.
7.
8.
Van Marter, L.J., Hernandez-Diaz, S., Werler, M.M., Louik, C. & Mitchell, A.A. Nonsteroidal antiinflammatory drugs in late pregnancy and persistent pulmonary hypertension of the
newborn. Pediatrics 131, 79-87 (2013).
Tyler, C.P. et al. Brain damage in preterm newborns and maternal medication: the ELGAN Study. Am. J. Obstet. Gynecol. 207, 192 e1-9 (2012).
Doyle, N.M., Gardner, M.O., Wells, L., Qualls, C. & Papile, L.A. Outcome of very low birth weight infants exposed to antenatal indomethacin for tocolysis. J. Perinatol. 25, 336-340
(2005).
Alano, M.A., Ngougmna, E., Ostrea, E.M., Jr. & Konduri, G.G. Analysis of nonsteroidal antiinflammatory drugs in meconium and its relation to persistent pulmonary hypertension of
the newborn. Pediatrics 107, 519-523 (2001).
Ojala, R. et al. Renal follow up of premature infants with and without perinatal indomethacin exposure. Arch. Dis. Child Fetal. Neonatal Ed. 84, F28-F33 (2001).
Ojala, R., Ikonen, S. & Tammela, O. Perinatal indomethacin treatment and neonatal complications in preterm infants. Eur. J. Pediatr. 159, 153-155 (2000).
Vermillion, S.T., Scardo, J.A., Lashus, A.G. & Wiles, H.B. The effect of indomethacin tocolysis on fetal ductus arteriosus constriction with advancing gestational age. Am. J. Obstet.
Gynecol. 177, 256-259; discussion 259-261 (1997).
Norton, M.E., Merrill, J., Cooper, B.A., Kuller, J.A. & Clyman, R.I. Neonatal complications after the administration of indomethacin for preterm labor. N. Engl. J. Med. 329, 1602-1607
(1993).
 0
0