Black Phosphorus Crystal Supplementary
advertisement

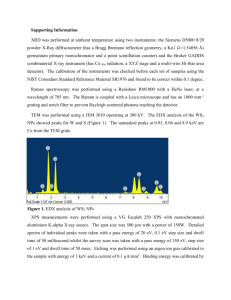
Supplemental Material for Manuscript Tunable photoluminescence from sheet-like black phosphorus crystal by electrochemical oxidation Z. X. Gan,1 L. L.Sun,1 X. L. Wu,1,* M. Meng,1 J. C. Shen,1and Paul K. Chu2,* 1 Key Laboratory of Modern Acoustics, MOE, Institute of Acoustics and Collaborative Innovation Center of Advanced Microstructures, National Laboratory of Solid State Microstructures, Nanjing University, Nanjing 210093, P. R. China 2 Department of Physics and Materials Science, City University of Hong Kong, Tat Chee Avenue, Kowloon, Hong Kong, China BP BP oxide Reflectance (%) 70 60 50 40 250 500 750 1000 1250 1500 Wavelength (nm) Figure S1. Diffusion reflectance spectra (DRS) acquired on the Shimadzu UV-3600 spectrometer with an integrated sphere attachment. The detector changes from a visible one to NIR at around 750 nm resulting in the sharp drop at 750 nm. that the bandgap does not change significantly. The similar DRS tend suggests Figure S2. DFT calculation conducted to theoretically confirm that the splitting of the 𝐴𝑔1 mode arises from surface oxidation. We first obtain structural optimization of the BP with surface dangling oxygen by calculating the total energy. (a) with the red ball representing oxygen. The optimized structure is shown in In the calculation, the plane wave basis and norm- conserving pseudo potentials with gradient exchange-correlation potential corrections are employed. Using the BFGS minimizer in the CASTEP package with default convergence tolerances of 1.0×10-5 eV for energy and 0.03 eV/A˚ for maximum displacement, the geometric structures of all the supercells are optimized to obtain stable structures or polymorphs with minimized total energy. Afterwards, the optimized geometrical structure is employed to construct and diagonalize the Hessian matrix as follows: ′ 𝑘,𝑘 𝐷𝛼𝛼 ′ = ′ 1 ′ √𝑀𝑘 𝑀𝑘 ′ 𝑘,𝑘 where Φ𝛼𝛼 ′ are the matrix force constants. 𝑘,𝑘 ⃗⃗⃗⃗𝛼 −𝑖𝑞⃗ .𝑟 ∑ Φ𝛼𝛼 ′ 𝑒 𝛼 The vibrational frequencies are obtained as the square roots of the eigenvalues belonging to each mode of the phonon wavevector 𝑞 = 0. The calculated Raman spectrum in (b) shows clear splitting in BP oxide compared to bulk BP. (a) A B C D Intensity (a.u.) 1200 800 400 0 100 200 300 400 500 -1 Raman shift (cm ) (b) 1400 Intensity (a.u.) 1200 1000 800 600 400 200 0 1000 2000 3000 4000 5000 6000 -1 Raman shift (cm ) Figure S3. (a) Raman spectra acquired from four different sheets after removing the oxidized surface layers. No Raman peaks appear below 300 cm-1 and the three sharp peaks corresponding to the 𝐴𝑔1 , 𝐵𝑔2 , and 𝐴𝑔2 modes confirm that the core of the BP oxide is not oxidized by the electrochemical treatment. (b) Long-range Raman spectrum showing no PL peaks indicating that oxidation occurs mainly occurs on the surface of BP oxide.