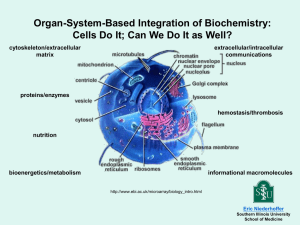
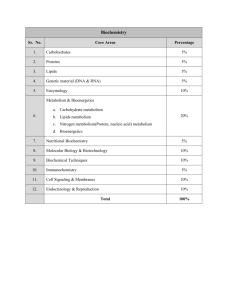
CHEM 222- Introduction to Biochemistry- Fall 2010
Instructor:
Didem Vardar-Ulu
Time and location: Tue,Wed&Fri 9:50-11:00 AM; SCI-220
Contact Info:
SCI-276, x3255 (office), SCI-301, X3285 (lab), dvardar@wellesley.edu
Office hours:
Tue: 2:30-4:00 pm, Fri: 11:00 – 12:30 pm (or by appointment)
Text: Biochemistry 4th edition: Reginald H. Garrett, Charles M. Grisham (Primary)
Lehninger Principles of Biochemistry, 4th edition: David L. Nelson, Michael M. Cox (Supplementary)
Biochemistry 6th edition: Jeremy M Berg, Lubert Stryer, John L. Tymoczko (Supplementary)
Course overview:
This course is designed as an introduction to the field of biochemistry. During this single
semester course we will focus on developing a strong conceptual understanding for the underlying basis of
a wide range of biochemical phenomena by revisiting many concepts that were covered in other chemistry
and biology courses. The emphasis will be in demonstrating how these two disciplines can be integrated
into a single interdisciplinary approach, to better understand and explain the world we live in. The course
is divided into three units: Fundamental building blocks of biochemistry, Enzyme Kinetics/Action, and
Medicinal Biochemistry/ Metabolism.
Biochemistry is an experimental science: So whenever possible, we will begin our discussions with
data drawn from the scientific literature and spend time to analyze and interpret experimental findings.
The Laboratory section will be an integral part of the course since essentially all of the material
discussed in lecture is the result of empirical biochemical research. Your lab experiments are designed to
help you appreciate the difficulties, limitations, and triumphs of “biochemistry at work”.
Biochemistry is a collaborative enterprise: So I will encourage and ask you to work in groups on
problems and exercises in and out of class. I have seen that group discussions are extremely helpful to
everyone in clarifying concepts that may be confusing when you study them on your own. Therefore,
there will also be optional weekly opportunities to have brainstorming sessions facilitated by an attached
tutor for the course. You are NEVER in competition with one another in my class. You are always
evaluated against an absolute standard of excellence.
Biochemistry is an interdisciplinary field: So, it will require you to master a new language to
describe certain familiar or not so familiar concepts. Becoming fluent in any language is challenging, and
takes different amounts of time and effort for each individual. However, it is very rewarding as it opens
up a whole new channel of communication.
Goals for the course:
1. To help you master enough conceptual understanding of biochemistry so that you can read
through, understand, and discuss a current problem in biochemistry at a fundamental level.
- Identify the main constituents of a given biological system and describe their physicochemical
properties.
- Establish a biochemically consistent relationship between the observed function of a given
biological system and the structure of its constituents.
- Use a multitude of available resources (published literature, online tools, current
instrumentation, etc.) to address and provide a plausible answer to a biochemical problem.
- Critically analyze a biochemical finding and evaluate the scientific validity of a biochemical
explanation.
2. To help you develop customized studying and learning techniques that would help you identify and
investigate any new problem in biochemistry.
1
Course conferences:
We will be testing out the new Sakai Platform this semester. I will be posting all the assignments
and any supplementary material on our Sakai Course site. We will also use this platform for any
announcements, so please check it out regularly.
Evaluation of work:
You will have different kinds of opportunities to demonstrate your learning throughout the
semester. Each work will be graded out of 100 and will be weighed according to the following rubric at
the end of the semester to determine the letter grade that reflects your overall performance in the
course.
1. Concept Checks (6 total, best 5 will be averaged): 10%,
2. Assignments: (Two activities and one problem set) 15%
3. Midterms (2): 15% each (Non-cumulative: scheduled for 10/26/10 and 12/1/10)
4. Final: 20% (self scheduled – comprehensive)
5. Laboratory: 25% (A passing grade of 50% in the lab is required to pass the course)
Extra Help:
I have included several suggested end of chapter questions (the detailed solutions manual for all
questions are at the library). I will also be posting OPTIONAL additional study questions (and answers)
for anyone interested in extra practice. I will be available during office hours and by appointment. I am
also happy to help out as much as I can via email. I also strongly encourage you to form study groups and
hold weekly brain storming sessions where you can discuss lecture and lab material. Based on past
experience, studying this material in groups greatly facilitates your learning. Finally, feel free to make
use of the tutoring services offered through PLTC or the chemistry help room.
Suggestions:
I highly value and appreciate any suggestion and constructive criticism you might have regarding
any aspect of this course. So please feel free to stop by or send me an email with any suggestions, any
time
Additional information:
Please let me know immediately if you foresee any conflict of important dates due to any personal
or religious reasons. I will be happy to consider making any necessary changes or accommodations in
advance.
Students with disabilities who are taking this course and who need disability-related
accommodations are encouraged to work with Verónica Darer, the Directory of Programs of the
Pforzheimer Learning and Teaching Center (for learning or attention disabilities), and Jim Wice, the
Director of Disability Services (for physical disabilities) to arrange these accommodations. Their offices
are in the Pforzheimer Learning and Teaching Center in Clapp Library.
2
CHEM222-F10 Lecture Schedule
Lecture
1
2
Date
09/07/10(T)
09/08/10 (W)
3
09/10/10(F)
4
09/14/10(T)
5
6
7
09/15/10 (W)
09/17/10(F)
09/21/10(T)
8
9
10
11
12
13
09/22/10 (W)
09/24/10(F)
09/28/10(T)
09/29/10 (W)
10/1/10(F)
10/5/10(T)
Topic
Getting to know each other and Introduction to Biochemistry
Review of fundamentals –
Intermolecular Interactions/ Acids and Bases/Thermodynamics
Amino acids, peptides
FUNDAMENTAL BUILDING BLOCKS OF BIOCHEMISTRY
Proteins I
Accessing Biochemical Information: Part 1: Literature search Workshop
IPT1 – Amino acids, peptides
Proteins II
Proteins III
Online biochemical information tools: Part 2: Bioinformatics Workshop
IPT2 – Proteins, understanding structure/ physicochemical basics
Proteins IV
Carbohydrates I
IPT 3– Proteins, understanding how structure relates to function
Carbohydrates II
Nucleic Acids I
14
15
16
17
18
19
10/6/10(W)
10/08/10(F)
10/12/10(T)
10/13/10(W)
10/15/10(F)
10/19/10(T)
10/20/10(W)
IPT4- Carbohydrates
Nucleic Acids II
(Monday schedule after Fall Break)
IPT5- Nucleic Acids
Lipids I
Lipids II (membranes)
IPT6 - Lipids
20
21
10/22/10(F)
10/26/10(T)
22
23
24
25
10/27/10(W)
10/29/10(F)
11/2/10(T)
11/3/10(W)
26
27
28
29
11/5/10(F)
11/9/10(T)
11/10/10(W)
11/12/10(F)
30
11/16/10(T)
Macromolecular Assemblies group presentations
MIDTERM I
ENZYMES
Tanner Conference
Enzyme kinetics I
Enzyme kinetics II
IPT 7– Understanding Enzyme Kinetics (Work on interpreting tables &
figures containing enzyme kinetics parameters kcat, Km, Vmax, etc.) Mona
Hall
Enzyme inhibition and regulation
Ligand binding proteins—myoglobin and hemoglobin
IPT 8– Enzyme Mechanisms Guest Lecturer: Prof. Dora Carrico-Moniz
Cooperativity and allostery in ligand binding proteins
MEDICINAL BIOCHEMISTRY/ METABOLISM
Bioenergetics
31
11/17/10(W)
32
11/19/10(F)
IPT 9– Guest Lecturer: Prof. Adele Wolfson (Thimet oligopeptidase) and
problems on coupled reactions/ bioenergetics
Bioenergetics/Introduction to metabolism
33
11/23/10(T)
Carbohydrate Metabolism I
34
11/24/10(W)
11/26/10(F)
No class
Thanksgiving
35
36
37
38
39
40
11/30/10(T)
12/1/10(W)
12/3/10(F)
12/7/10(T)
12/8/10(W)
12/10/10(F)
Self Scheduled
* This Chapter is from
Reading
Ch 1-2
Ch 1-2
Ch 4
4, 5, 6, 7, 11, 13, 16, 17
Ch 5
1, 5, 8 (use Table 5.2), 14,
16
Ch 6
1, 2, 5, 7, 9, 13, 14, 15
Ch 7
2, 3, 4, 6, 8, 12, 19, 20
Ch 7
Ch 10
Ch 11
3
Activity 1 due
1, 2, 4, 5, 6, 18
DNA movie night
3, 4, 5, 8, 10, 12, 13, 19
Ch 8
Ch 8-9
Ch8: 1, 2, 10, 12, 17, 19,
20, 22
Ch9: 1, 2, 4, 5, 7, 13, 14,
17, 23
Activity 2 due
Ch 13
Ch 13,15
Ch 13: 1, 2, 4, 5, 10, 12, 16
Ch 15: 2
Ch 13, 15
Ch 5.1*
Ch. 14
Ch 5.1*
Ch 14: 1, 2a-b
Ch 15: 5, 7, 13, 14
Ch 3
Ch 3
Ch 3 , 17
Ch 18
Carbohydrate Metabolism II
MIDTERM II
Basics of Metabolic Regulation
Tricarboxylic Acid Cycle
Electron Transport Chain and Oxidative Phosphorylation
Discussion on Medical Myths/ Literature examples; Q&A
Final Exam
“Lehninger Principles of Biochemistry” Cox Nelson, 2008
Chapter Problems
Ch1: 1,3,5,16,17
Ch2: 1-9, 11, 14, 10, 12,
13, 17, 19, 21, 24
Ch22
Ch 15 *
Ch 19
Ch 20
Ch3: 1, 2, 3, 5, 6, 9,
12, 14, 17, 20
Ch 17: 4, 6, 7, 8
Problem Set due
Ch 18: 1, 8, 9, 10, 12, 13,
22
Ch 22: 2, 4, 7, 8
 0
0