Periodic Table
advertisement

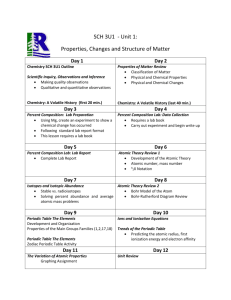
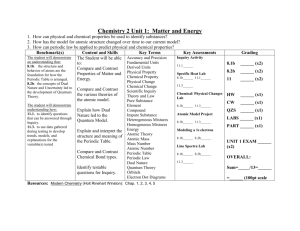
Name ______________________________ Class ______ Date ___________________ Periodic Table Scavenger Hunt Use your science textbook and an Internet search engine such as Google to answer the following questions. 1. What is a chemical symbol? 2. Why do some elements have symbols that don’t use letters in the element’s name? 3. Fill in the chemical symbols for the following elements. NAME SYMBOL NAME Tungsten Silver Potassium Mercury Lead Gold SYMBOL 4. Differentiate between atomic number and atomic mass. 5. Look up the following elements and list their atomic number and their atomic mass. Name Atomic Number Atomic Mass Name Gold Hydrogen Mercury Xenon Bromine Iodine Neodymium Platinum Potassium Radon Atomic Number Atomic Mass 6. What state of matter are most of the elements in the periodic table? __________________ 7. What are the only two elements that are liquids at room temperature? 8. Why are Nobel Gases sometimes called the ”Inert Gases”? 9. Complete the chart below. The Periodic Table trends Describe what happens to reactivity of elements as you move left to right across the periodic table. Describe what happens to atomic mass as you move left to right across the periodic table. Describe what happens to atomic mass as you move top to bottom across the periodic table. Describe what happens to atomic number as you move left to right across the periodic table. Describe what happens to atomic number as you move top to bottom across the periodic table. 10. What feature on the Periodic Table separates the metals and nonmetals? 11. Some elements are radioactive. What does radioactive mean? Name 3 radioactive elements. 12. Complete the following rable. The Transition METALS Define transition metals. Where are they on the periodic table? List 3 physical properties of transition metals. List 3 examples. The Metalloid Family Define metalloid. List 3 physical properties of metalloids. The Halogen Family Halogen = from the Greek words meaning... Interesting Fact Halogens that are SOLID They are the only family to have a solid, liquid, AND gases in their family. Halogens that are LIQID Halogens that are GASES The Alkali Family List the elements that make up this family. What happens when you combine these elements with water? (Read the caption for figure 9 on page 321.) What are the physical properties of alkali metals? The Alkaline List the elements that make up this family. Describe the physical properties of alkaline earth metals. (Last 2 sentences on page 321.) Earth Family