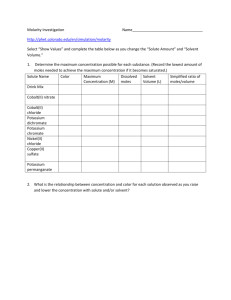
For Students
Engineering Design in Oregon Science Classrooms
Page 1 of 3
Calorimeter Reading Handout
Do you ever wonder exactly how it is known how
many calories are in a delicious bag of chips? Or how much
energy is in a refreshing can of pop? What about chemical
reactions like combustion of hydrogen and oxygen? It
reacts so quickly, how can a scientist or engineer even
begin to measure the energy released? How can we scale
reactions for different sizes and amounts of the reactants?
Many of you might be thinking "Well, they look it up of
course!" Ultimately, the data had to come from somewhere.
The answer to all these questions is calorimetry. Calories
are a measure of energy including chemical energy. If we
react our item of interest in the presence of a substance
with a known heat capacity, we can measure the
temperature change of the known substance using a
calorimeter. Oftentimes, this substance is water. We can
then relate the temperature change to the amount of heat
energy transferred using equations of heat transfer.
Figure 1: The DZero Liquid Argon Calorimeter is used
to detect subatomic particles in complex quantum
mechanical experiments.
Source: http://bit.ly/19Qf3RC
A calorimeter is a fairly simple device. All that is required is a chamber in which to house an energy
generating reaction, and some substance with a known heat capacity to capture the heat energy generated. There
are many different types of calorimeters. For example, in large particle accelerators, physicists use specialized
calorimeters to detect the quantized energy given off by the presence of rare subatomic particles. Food chemists
use bomb calorimeters to measure the energy content of food. A bomb
calorimeter is a type of constant volume calorimeter which is divided into
two parts. Imagine a cylinder within a cylinder. The smaller cylinder, which
is made of a highly conductive material, contains an excess of oxygen, an
electrical igniter, and the food in question. The larger cylinder surrounding it
contains water and a thermometer and is well insulated.
Figure 2: A Sample Calorimeter
Source: http://bit.ly/19Qf3RC
For this activity, we will use water because it is readily available and
its properties well understood. Depending on the type of calorimeter used,
this can be housed in the same chamber as the reaction (such as dissolving
one substance in another) or in a different chamber (such as for a
combustion reaction). Heat transfer to the surrounding structure of the
calorimeter should be minimal. The heat energy should instead go into the
measuring substance to raise its temperature. Where do you think the
calorimeter would lose heat energy? For simplicity’s sake, we will assume
that the moving water is heated uniformly when stirred effectively.
A much simpler calorimeter is a coffee cup calorimeter. Though there are variations, this works on the
same principle as other calorimeters. In this case, a cup holds the reaction of interest, and a thermometer
measures the temperature change. In this activity, the dissolution of potassium chloride salt (KCl) in water is the
reaction of interest. We want to maximize the amount of energy going into the water, so the cup should be as
insulating as possible. Multiple layers might even be used (that is, a cup nested inside of another cup). We also
For Students
Engineering Design in Oregon Science Classrooms
Page 2 of 3
want to minimize losses elsewhere. Where else might we lose heat energy? The mechanism of transport of heat
energy in this device includes both conduction through the barrier and convection of the water. In order for our
assumption of uniform temperature to be valid, it must be stirred, no matter the design. Why does this make a
difference? Well, stirring the water helps ensure that the hot and cold water in the cup are well mixed. This
assures us that the heat will be evenly distributed and that we will get an accurate temperature reading. If we
have large heat energy losses out of our cup calorimeter we will not get an accurate measurement of how much
energy our reaction produces or requires.
Measuring Heat Transfer with Our Calorimeter
In order to figure out how much energy is produced using our calorimeter, we need to use the formula
for heat transfer (Q). Heat transfer is related to the change in temperature change (ΔT), the heat capacity (Cp)
of the material and the mass of the material. The equation is as follows:
𝑄 = 𝑚𝐶𝑃 ∆𝑇
Because the values we calculate using this formula are based on experimental measurements, it is
sometimes referred to as Qexp
In this case, we want to use the heat capacity, mass and temperature change of the water.
The heat capacity of water, 𝐶𝑃 = 4.186 J/(g·C)
Sample calculation.
200 ml of water is heated by 20° C. How much heat was transferred?
We’ll use Qexp = 𝑚𝐶𝑃 ∆𝑇
The mass of 200 ml of water is 200 g. The change of temperature, ∆𝑇, is 20 ° C.
We also know that heat capacity of water, 𝐶𝑃 = 4.186 J/(g·C)
So, Qexp = 𝑚𝐶𝑃 ∆𝑇= 200 g x 4.186 J/(g·C) x 20 ° C = 16,744 J or 16.7 kJ.
Predicting the Energy Used to Dissolve Potassium Chloride
We’re also interested in predicting the amount of energy required to dissolve potassium chloride in
water. The heat of solution is given by
𝑄 = −∆𝐻𝑛
where n is the number of moles of our potassium chloride salt and ∆H is the change in enthalpy per mole
of our system. Enthalpy is a special measure of energy in a system. In this case, it is the energy required to
dissolve our salt. In the case of potassium chloride the dissolution reaction is considered endothermic because
it uses or absorbs energy. Endothermic dissolutions have positive change-in-enthalpy values. Dissolutions that
produce energy like potassium hydroxide are called exothermic and have negative change-in-enthalpy values.
To change heat of solution equation to use mass rather than moles we again use
𝑛 =
𝑚𝑎𝑠𝑠
𝑚𝑜𝑙𝑎𝑟 𝑚𝑎𝑠𝑠
The molar mass for potassium chloride is 74.6 grams per mole.
The enthalpy of dissolution, ΔH, for potassium chloride is 17.2 kJ/mol.
For Students
Engineering Design in Oregon Science Classrooms
Page 3 of 3
Because this predicted value for Q is based on theory and accepted values for potassium it is sometimes referred
to as Qideal
Sample calculation
How much energy is required to dissolve 10.0 grams of potassium chloride in water?
We use the formula for heat of solution: 𝑄 = −∆𝐻𝑛
We know that ΔH for potassium chloride is 17.2 kJ/mol.
We need the number of moles of potassium so we use 𝑛 =
𝑚𝑎𝑠𝑠
𝑚𝑜𝑙𝑎𝑟 𝑚𝑎𝑠𝑠
We know the molar mass of potassium chloride is 74.6 grams per mole and we are dissolving 10 grams
so the number of moles we dissolving is n = 10.0 / 74.6 = 0.134 moles.
So, the heat associated with dissolving 10 grams of potassium chloride is. It is a negative number
because heat is absorbed as dissolving potassium chloride is endothermic.
Qideal = −∆𝐻𝑛 = -17.2 kJ/mol x 0.134 moles = -2.31 kJ
 0
0