stoichiometry and titration practice
advertisement

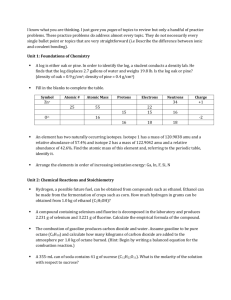
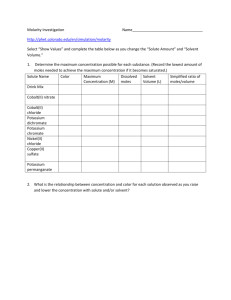
1) Hydrazine, N2H2, reacts with hydrogen peroxide, H2O2, to produce nitrogen gas and water. How many moles of hydrazine are required to produce 8.6 moles of water? 2) If 8.5 moles of calcium carbide react with an excess of water, how many moles of acetylene (C2H2) are produced? CaC2 (s) + 2H2O (l) → Ca(OH)2 (aq) + C2H2 (g) 3) How many grams of oxygen are required for the complete combustion of 37.06g of butane, C4H10? 3) Potassium chloride can be produced from its elements: K (s) + Cl2 (g) → KCl (s) If 35g of potassium are combined with 15g of chlorine, how much potassium chloride is produced? How much potassium and how much chlorine are left when the reaction is complete? 4) Hydrochloric acid reacts with calcium carbonate to produce calcium chloride, carbon dioxide, and water. HCl (aq) + CaCO3 (s) → CaCl2 (aq) + H2O (l) + CO2 (g) How many milliliters of a 6.0 M HCl solution are required dissolve 22.348g of calcium carbonate? 5) Aspirin (C9H8O4) is produced from the reaction of acetic anhydride (C4H6O3) with salicylic acid (C7H6O3): C7H6O3 (s) + C4H6O3 (l) → C9H8O4 (s) + C2H4O2 (l) a. How much aspirin can theoretically be produced from 3.00g of salicylic acid and 5.40g of acetic anhydride? b. What is the percent yield if 2.04 grams of aspirin are produced? 6) Dr. Unfried recently prepared a hydrochloric acid solution in the lab. He determined the acid concentration by titration using a 0.12M sodium hydroxide solution. It took 36.2mL of the NaOH solution to neutralize (faint pink endpoint) 24.6mL of the acid solution. What is the concentration of the HCl solution? 7) How many milliliters of a 0.60M sulfuric acid, H2SO4, solution would be required to react with (neutralize) 23.0g of sodium carbonate? 8) The diprotic acid, oxalic acid (H2C2O4) reacts with potassium permanganate as follows: + -2 - +2 16H + 5C2O4 + 2MnO4 → 10CO2 + 2Mn + 8H2O How many milliliters of a 0.0100 M solution of potassium permanganate solution would be needed to react with 1.50g of oxalic acid? 9) (Varsity!) Dr. Unfried recently prepared a nitric acid solution in the lab. He standardized the acid solution by titrating with a sodium hydroxide solution, and standardized the sodium hydroxide solution with potassium acid phthalate (KHP). It took 36.2mL of the NaOH solution to neutralize 34.9mL of the HNO3 and took 28.4mL of the NaOH solution to reach a pink endpoint with 0.5068g of KHP. The molar mass of KHP is 204.22g/mol. What is the concentration of the HNO3?