Handout
advertisement

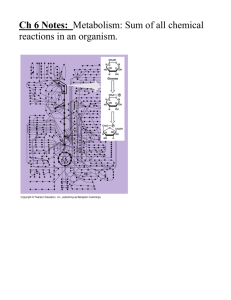
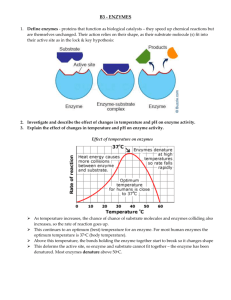
2.4 Chemical Reactions Notes Key Concept: Life depends on chemical reactions. Name ______________________________ Date _____________ Period ___________ Bonds break and form during chemical reactions. -Chemical reactions chance substances into different once by breaking and forming chemical bonds. -_____________________________________ during a chemical reaction. - ____________________________________ by a chemical reaction. Chemical reactions release or absorb energy. -Activation energy is the amount of energy that needs to be absorbed to start a ___________________. Exothermic reactions release more energy than they absorb. -Reactants have higher bond energies than products. -Excess excess energy _____________________________ by the reaction. -_____________________________ the reaction. (Exo = Exit) Endothermic reactions absorb more energy than they release. - Reactants have lower bond energies than products. -____________________________________ by the reaction to make up the difference. -_____________________________ the reaction. (Endo= “into”) 2.5 Enzymes Key Concept: Enzymes are catalysts for chemical reactions in living things. A catalyst lowers activation energy. -Catalysts are substances that ______________________________________________________ -Decrease activation energy*** -Increase reaction rate*** Enzymes allow chemical reactions to occur under tightly controlled conditions. -Enzymes are ___________________________________________________. -Enzymes are needed for almost all processes. -Most ___________________________________________________. An enzyme’s structure allows only certain reactants to bind to the enzyme. -Substrate: ________________________________________________________________________ -Active site: _______________________________________________________________________ Lock and key model shows how enzymes function -Substrates are ________________________________________________ of the enzyme. -Enzyme ________________________________________________________________. -Reaction forms a product that is _____________________________________________. -Enzyme remains unchanged and can be used again & again. Disruptions in homeostasis can prevent enzymes from functioning. -Enzymes function best in a small range of conditions. -__________________________________________________ can break hydrogen bonds. - An enzyme’s _________________________________________________________. Concentration of enzyme & substrate can also affect enzyme function. -Enzyme concentration: If the concentration of the substrate is kept constant and the concentration of the enzyme is increased, the rate of reaction increases linearly. -If the concentration of enzyme is _____________, the reaction rate _______________. -Substrate concentration: If the concentration of the enzyme is kept constant and the concentration of the substrate is increased, initially, the rate increases with substrate concentration, until the rate levels out and remains __________________.