Chapter 12 Solutions: Chemistry Reading Guide
advertisement

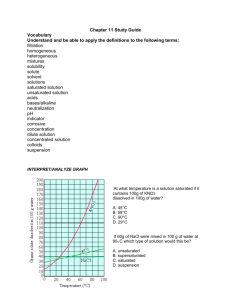
CP Chemistry Mrs. Klingaman Chapter 12: Solutions Name: ___________________________________________ Mods: __________________ Chapter 12: Solutions Reading Guide 12.1 – Types of Mixtures (pgs. 379-384) 1. Define soluble – 2. Define solution – 3. Define solvent – 4. Define solute – 5. The solute is designated as the component of a solution that is of ________________ quantity. 6. Solutions may exist as _________________, __________________, or _________________. Fill in Figure 1.3 which shows some possible solute-solvent combinations for solutions. Solute state Solvent state Example 7. Give an example of an alloy and list the properties of that alloy that make it more desirable than the individual pure metals from which it is composed. 8. Define suspension – 9. Define colloid – a) An _________________ is a liquid in a liquid example of a colloid, like milk. b) Clouds and fog are examples of colloids called _________________ ______________ in which a liquid is dispersed in a gas. c) _____________ is a type of colloid in which a gas is dispersed throughout a liquid. d) A __________ is a type of colloid in which a solid is dispersed throughout a liquid. 10. Define Tyndall effect – The Tyndall effect can be used to distinguish a _______________ from a ________________. 11. Define Brownian motion – 12. Fill in Figure 1.7 which shows the properties of solutions, colloids, and suspensions. Solutions Colloids Suspensions 13. Define electrolyte – 14. Define nonelectrolyte - 12.2 – The Solution Process (pgs. 385-395) 15. Name the three factors that affect dissolving and describe how each factor speeds up the dissolving process. 16. Define solution equilibrium – 17. Define saturated solution – 18. Define unsaturated solution – 19. Define supersaturated solution – 20. Define solubility - 21. What do we mean by the term “like dissolves like”? 22. Explain how crystals of lithium chloride dissolve in water. 23. The solution process with water as the solvent is referred to as ___________________. 24. Define immiscible – 25. Define miscible – 26. Changes in pressure have very little effect on the solubilities of _______________ and ______________ in liquid solvents. However, increases in pressure _______________ gas solubilities in liquids. 27. Henry’s Law states: 28. Define effervescence – 29. For a gas: increasing the temperature usually __________________ the solubility. 30. For a solid: increasing the temperature ___________________ the solubility. However, an ______________________ temperature increase can result in a _______________ increase in solubility for some solvents and only a ________________ change for others. 31. Solution formation occurs in three steps according to Figure 2.12. Step 1: Bonds between solute particles break; energy is ____________________. Step 2: Bonds between solvent particles break; energy is ____________________. Step 3: Bonds between solute and solvent particles form; energy is ___________________. ∆Hsolution = ∆HStep1 + ∆HStep2 + ∆HStep3 If ∆Hsolution has a negative value, the reaction is ______________________________. If ∆Hsolution has a positive value, the reaction is _______________________________. 12.3 – Concentration of Solutions (pgs. 396-403) 32. Define concentration – 33. What is the difference between a dilute solution and a concentrated solution? 34. Define molarity (M) and give the formula for calculating molarity – 35. Define molality (m) and give the formula for calculating molality – Factors that Affect Solubility: In-Class Notes Solids and Liquids- There are four factors that affect the solubility of solids and liquids. They are: 1) 2) 3) 4) *Changes in pressure have such a minimal effect on the solubility of a solid or liquid in solution that they are considered negligible. Gases- There are three factors that affect the solubility of a gas. They are: 1) 2) 3) . Intro to Solubility Curves & Types of Solutions 1. Define solubility- 2. For most substances, solubility increases as temperature increases. What substances are the exceptions on the graph below? ________________________ Part One: Reading Solubility Curves Use the graph to answer the following questions. REMEMBER UNITS! 3. What mass of solute will dissolve in 100 mL of water at the following temperatures? a) KNO3 at 70°C _______________ b) NaCl at 100°C _______________ c) NH4Cl at 90°C _______________ d) Which of the above three substances is most soluble in water at 15°C. ________________ 4. What mass of solute will dissolve in 200 mL of water at the following temperatures? a. KNO3 at 70°C ____________ b. NaCl at 100°C ____________ c. NH4Cl at 90°C ____________ Part Two: Types of Solutions (saturated, unsaturated, supersaturated) On a solubility curve, the lines indicate the concentration of a saturated solution = contains the _____________________ amount of solute that will dissolve at that specific temperature (no additional solute can be dissolved in these solutions at that temperature. If any more solid is added, it will remain _____________________ at the bottom of the beaker) Values on the graph _______________ a curve represent unsaturated solutions = more solute could still be dissolved into the solution at that temperature Values on the graph _______________ a curve represent supersaturated solutions = contains _______________ than the maximum amount of solute that can be dissolved at that temperature (HOW?!? - heat the water, add a LOT of solute and slowly ______________ the solution so that at the lower temperature the solution contains more solute than it should be able to) Solubility & Solutions Practice Directions: Use the solubility curve on the previous page to label the following solutions as saturated or unsaturated. If unsaturated, write how much more solute (grams) can be dissolved in the solution. Solution Saturated or Unsaturated? If unsaturated: How much more solute can dissolve in the solution? a solution that contains 70g of NaNO3 at 30°C (in 100 mL H2O) a solution that contains 50g of NH4Cl at 50°C (in 100 mL H2O) a solution that contains 20g of KClO3 at 50°C (in 100 mL H2O) a solution that contains 70g of KI at 0°C (in 100 mL H2O) Additional Practice: 1. Use the solubility chart on the previous page to answer the following questions: a. At 90C, you dissolved 10 g of KCl in 100. g of water. Is this solution saturated or unsaturated? _________________________________. How do you know? ________________________________________________________________ b. A mass of 100 g of NaNO3 is dissolved in 100 g of water at 80ºC. Is the solution saturated or unsaturated?_______________________________________. As this solution is cooled, at what temperature will a precipitate (solid) form? ___________ c. Which compound is most soluble at 20 ºC? _________________ d. Which compound is the least soluble at 40 ºC? _________________ e. Which substance on the chart is least soluble at 10C? ________________ f. A mass of 80 g of KNO3 is dissolved in 100 g of water at 50 ºC. This solution is then heated to 70ºC. How many more grams of potassium nitrate must be added to make the solution saturated at this new, warmer temperature? Explain your reasoning. Solubility Curve Practice Worksheet Use the solubility chart below to answer the following questions: Graph from U. Va Department of Physics. 1) What is the solubility of potassium nitrate at 300 C? 2) How many grams of ammonia can I dissolve in 200 grams of water at a temperature of 450 C? __________________ __________________ 3) At what temperature is the solubility of sodium chloride the same as the solubility of potassium chloride? __________________ 4) How many grams of ammonium chloride would I need to make 300 grams of a saturated solution at 700 C? __________________ 5) What do all of the compounds that decreased in solubility over the temperature range in the graph have in common? __________________ 6) What compound is least soluble at 400 C? __________________ 7) What ionic compound is least soluble at 400 C? __________________ Measuring Concentration - Molarity and Molality Important Equations: Molarity (M) Example Problems: 1) Calculate the molarity of a solution made by dissolving 0.0278 moles of glucose in enough water to form 0.15 L of solution. 2) Calculate the molarity of a solution made by dissolving 23.4 grams of Na2SO4 in enough water to form 125 mL of solution. Molality (m) Example Problems: 1) What is the molality of a solution when 0.28 moles of C10H8 is dissolved in 0.425 kg of C7H8? 2) Find the molality of a solution when 4.75 g of HCl is dissolved in 64 g of water. Molarity (M) Practice Problems 1) Calculate the molarity when 0.50 moles of MgCl2 are dissolved in 1.5 L of solution. 2) Calculate the molarity when 1.24 moles of KCl are dissolved in 750 mL of solution. 3) Sea water contains roughly 28.0 g of NaCl for every 1 L of solution. What is the molarity of sea water? 4) Calculate the moles of solute in 1250 mL of a 0.64 M NaCl solution. 5) What volume (in mL) of 12.0 M HCl is needed to contain 3.0 moles of HCl? Molality (m) Practice Problems 1) If 3.4 moles of Na2CO3 are dissolved in 1.74 kg of water, what is the molality of this solution? 2) What is the molality of a solution that consists of 56.7 g of SrCl2 dissolved in 1.325 kg of water? 3) What is the molality of a solution that consists of 0.20 moles of FeCl3 dissolved in 2250 g of water? 4) How many moles of solute are present in a 1.15 m solution that contains 0.096 kg of solvent? 5) How many kilograms of solvent are present in a 0.038 m solution that contains 0.019 moles of solute? Colligative Properties 1. Define colligative properties – 2. What are the four Colligative Properties? (1) _______________________________________________________ (2) _______________________________________________________ Example: (3) _______________________________________________________ Example: (4) _______________________________________________________ 3. Define osmosis and include a picture – 4. Define hypertonic & include a picture – 5. Define hypotonic & include a picture – Solutions Review Worksheet Part 1: Solubility Curves Directions: Answer each of the questions below using the solubility curves provided. 1. Which of the substances are gases? How do you know? 2. How many grams of Sodium Nitrate can be dissolved in 200 grams of water at 50ºC? 3. At 60ºC, 70 grams of ammonium chloride is added to 100 grams of water. Is the solution, saturated, unsaturated or supersaturated? 4. A solution is made with 40 grams of NaCl and 100 grams of water at 80ºC and then the solution is cooled down to 20ºC . The excess NaCl stays in solution. Is the solution saturated, unsaturated or supersaturated? Part 2: Solvation 1. What is solvation? 2. Complete describe how sodium chloride dissolves in water. Use pictures, if necessary. Part 3: Molarity 1. What is the equation for Molarity (M)? 2. A 800 mL salt solution contains 0.7 moles of NaCl. What is the molarity of the solution? 3. How many grams of table sugar (C12H22O11) will be needed to make 3.5 liters of a 1.15 M solution? 4. If you have 188 grams of CaCl2 and 2.6 liters of solution, what will the molarity of the solution be? 5. How many liters of solution would you need if you wanted to make a 6.0 M if all you had was 50.0 grams of AlBr3? Part 4: Molality 1. What is the equation for molality (m)? 2. What is the molality of a solution that contains 63.0 g HNO3 in 0.500 kg H2O? 3. What is the molality of a solution that contains .500 mol HC2H3O2 in 125 g H2O? 4. What mass of water is required to dissolve 100 g NaCl to prepare a 1.50 m solution? 5. What mass of water must be used to dissolve 0.500 kg C2H5OH to prepare a 3.00 m solution?