Intro to Aqueous Solutions Notes
advertisement

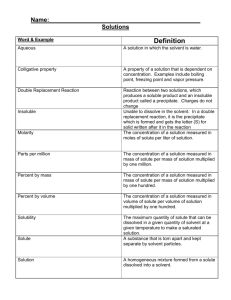
Introduction to Aqueous Solutions Notes Most interesting chemical reactions happen in aqueous solutions. Review how a solutions are formed. (A solute is dissolved in a solvent.) Review that both solutions have a concentration, which is a numeric reflection of the moles of solute compared to the volume of solution (M or moles/litre). It may also be necessary to review stoichiometry. A solution may be diluted using the formula: M1V1 = M2 V2,, which assumes that the moles of solute remain constant while the volume of solution is changing. When two aqueous solutions are mixed together, a precipitate reaction may or may not occur. The precipitate produced by the reaction is insoluble, or slightly soluble, in the aqueous product. In order to predict if a precipate reaction will occur, solubility rules are referenced. Solubility: is understood as a maximum amount of solute that dissolves in a solvent at so called equilibrium. In chemistry, an equilibrium is a state where reactants and products reach a balance - no more solute can be dissolved in the solvent in the constant conditions (i.e., temperature, pressure). Such a solution is called a saturated solution. Precipitation: is the formation of a solid in a solution or inside another solid during a chemical reaction or by diffusion in a solid. When the reaction occurs in a liquid, the solid formed is called the precipitate, or when compacted by a centrifuge, a pellet. The liquid remaining above the solid is in either case called the supernate or supernatant. Example 1: NaCl(s) dissolved in water Simulation: http://phet.colorado.edu/en/simulation/soluble-salts Molecular level (diagram): Symbolic level (formula): Example 2: NaCl(aq) + AgNo3(aq) combined together Go to simulation for precipitation (double displacement) reaction between silver nitrate and sodium chloride: http://www.crescent.edu.sg/crezlab/Webpages/PptReaction4.htm# Molecular Level: Symbolic Level: