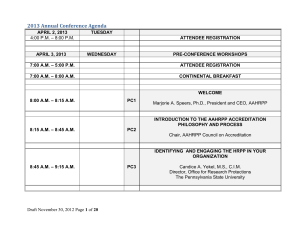
Requirements for AAHRPP Accreditation

IRB: Requirements for AAHRPP Accreditation
Policy number: 900 Date: 06/11/2014
References:
Maintaining Accreditation : http://www.aahrpp.org/apply/maintainingaccreditation/applying-for-reaccreditation
Cross Reference:
101 A Role of the Institutional Official
600 HRPP Components
901 Periodic Evaluation
Policy Owner:
Executive Director, HRPP
Definitions:
None
1.0 Reason for Policy
To describe accreditation of the Human Research Protection Program for the University of Minnesota and its healthcare component institutions.
2.0 Scope of Policy
This policy covers the University of Minnesota and its healthcare components.
3.0 Policy Statement
The Human Research Protection Program (HRPP) of the University of Minnesota applies for accreditation from the Association for the Accreditation of Human Research Protection Programs
(AAHRPP). It received initial Qualified Accreditation in 2004 and then Full Accreditation in 2005, which was renewed in 2007 and 2010.
Applying for Accreditation and Renewal
According to the policies set forth by AAHRPP, Accredited Organizations must renew their accreditations three years after the initial accreditation and every five years thereafter. Instructions for the application are found at the AAHRPP web site http://aahrpp.org/ .
Typically, a team of senior staff members in the HRPP will conduct a self-assessment of the program, with input from components of the HRPP. The staff then completes the requirements for and submits
Page 1 of 2
the initial application for re-accreditation, following sign off and approval from the Institutional Official.
This application must be submitted at least 12 months in advance of the AAHRPP Council of
Accreditation meeting, which is assigned based on the date of the previous accreditation.
Additional AAHRPP Requirements
AAHRPP requires annual reports of activities and payment of annual fees to maintain accreditation.
In addition to the annual report, AAHRPP must be notified as soon as possible, preferably within 72 hours of any:
Inquiries from a governmental oversight office, such as the OHRP or the FDA when the inquiry could result in a for cause investigation.
Any findings or changes concerning the HRPP that might affect the program’s ability to continue to meet the AAHRPP standards.
The HRPP must report within 24 hours, any sanctions taken by a government oversight office.
If there is any doubt about making a report, the HRPP should contact the AAHRPP office for direction.
As with any reporting action, the HRPP relies on the Institutional Official to formally transmit written notice of reports.
Failure to follow all AAHRPP requirements pertaining to accreditation/re-accreditation and reporting may result in revocation of accreditation status.
4.0 Required approvals for this document
Title
Executive Director, HRPP
5.0 Revision History
Revision Reason for change
06/01/14 Update
01/11/11 New Policy
Date of release
09/02/14
01/11/11
To obtain a copy of a historical policy, e-mail IRB at irb@umn.edu or call 612-626-5654
Page 2 of 2