File - Heidi Stevenson`s Portfolio
advertisement
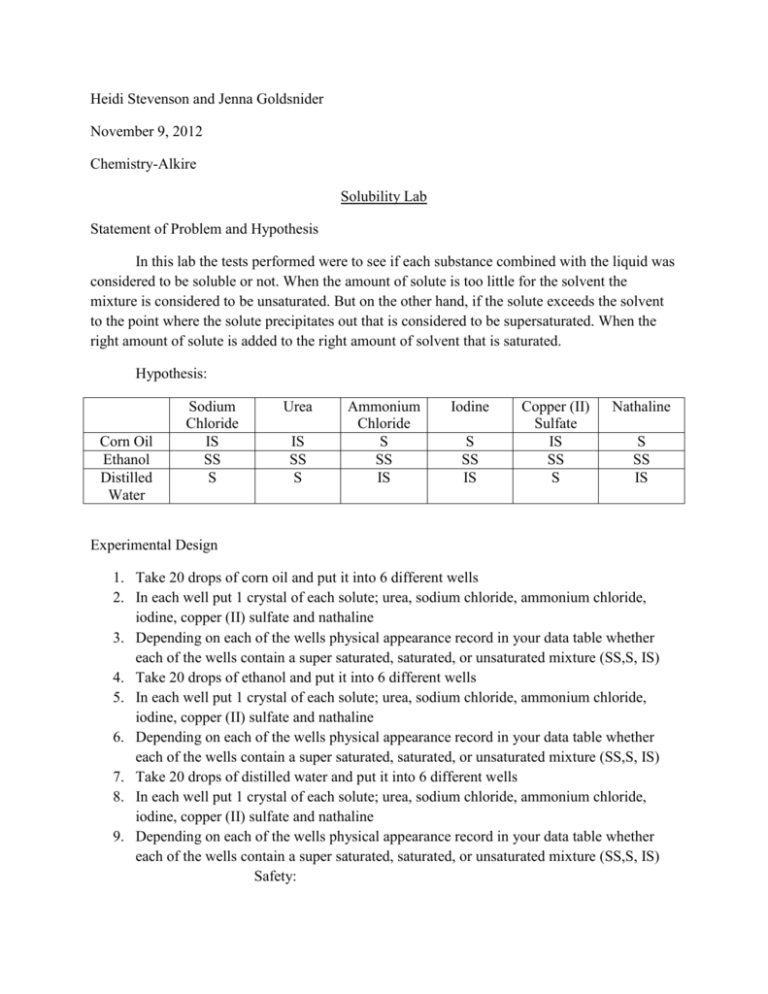
Heidi Stevenson and Jenna Goldsnider November 9, 2012 Chemistry-Alkire Solubility Lab Statement of Problem and Hypothesis In this lab the tests performed were to see if each substance combined with the liquid was considered to be soluble or not. When the amount of solute is too little for the solvent the mixture is considered to be unsaturated. But on the other hand, if the solute exceeds the solvent to the point where the solute precipitates out that is considered to be supersaturated. When the right amount of solute is added to the right amount of solvent that is saturated. Hypothesis: Corn Oil Ethanol Distilled Water Sodium Chloride IS SS S Urea IS SS S Ammonium Chloride S SS IS Iodine S SS IS Copper (II) Sulfate IS SS S Nathaline S SS IS Experimental Design 1. Take 20 drops of corn oil and put it into 6 different wells 2. In each well put 1 crystal of each solute; urea, sodium chloride, ammonium chloride, iodine, copper (II) sulfate and nathaline 3. Depending on each of the wells physical appearance record in your data table whether each of the wells contain a super saturated, saturated, or unsaturated mixture (SS,S, IS) 4. Take 20 drops of ethanol and put it into 6 different wells 5. In each well put 1 crystal of each solute; urea, sodium chloride, ammonium chloride, iodine, copper (II) sulfate and nathaline 6. Depending on each of the wells physical appearance record in your data table whether each of the wells contain a super saturated, saturated, or unsaturated mixture (SS,S, IS) 7. Take 20 drops of distilled water and put it into 6 different wells 8. In each well put 1 crystal of each solute; urea, sodium chloride, ammonium chloride, iodine, copper (II) sulfate and nathaline 9. Depending on each of the wells physical appearance record in your data table whether each of the wells contain a super saturated, saturated, or unsaturated mixture (SS,S, IS) Safety: 1. 2. 3. 4. 5. 6. 7. Wear goggles at all times during the lab. Use a lab apron to cover up the clothes you are wearing. Do not have baggy sleeves during the experiment Have your hair tied back if it is should length or longer You must wear closed toed shoes in the lab area Be aware of all exits out of the room in case of fire Do not cross contaminate any solutions Data and Observation Corn Oil Ethanol Distilled Water Sodium Chloride IS IS S Urea IS IS S Ammonium Chloride IS IS IS Iodine S SS IS Copper (II) Sulfate IS IS IS Nathaline IS SS IS Conclusion Our hypothesis in the end was proven to have its faults but was overall correct. We mainly did not have an understanding on ethanol since we thought it was going to be slightly soluble no matter what instead of insoluble on occasions. Our data can be considered reliable considering there was no cross contamination or other tampering that could result in unreliable results. Improvements to the experiment could include testing each solute twice to confirm the accuracy when dealing with the solvents. Heidi Stevenson Questions: 1. Corn oil is not very likely to be dissolved in the river because like dissolves like and an oil does not mix well with water. 2. While glancing over some of the lab partners around our lab station I came to the conclusion that most of our answers if not all were matched up for the most part. 3 a. When the solute that is placed in water is considered to be soluble in most cases the same solute placed in ethanol would be considered to be insoluble, and vice versa. b. Both lamp oil and ethanol serve a purpose for heat combustion and are both nonpolar. 4 a. There were 7 solubility observations that we found were to be unexpected. b. For the hypothesis we thought that corn oil and Ammonium Chloride as well as Napthaline would be soluble but in reality bother were considered to be insoluble. For ethanol when mixed with Sodium Chloride, Urea, Ammonium Chloride, and Copper (II) Sulfate we predicted that the substance would be considered to slightly soluble. After the lab was completed we realized that instead of being slightly soluble they were insoluble. When distilled water and Copper (II) Sulfate are mixed we thought it would be soluble when it really was insoluble. 5. All of the ethanol based mixtures were either slightly soluble or insoluble, there was no just plain soluble. With corn oil as well as distilled water it was always insoluble or soluble, never considered to be slightly soluble. 6 a. Hexane b. 7 Ethylene Glycol a. Sodium Chloride Ionic (not a covalent bond) Sodium Chloride S Sodium Chloride IS Urea Polar (soluble in water) Urea S Urea IS Ammonium Chloride IS Iodine IS Ammonium Chloride IS Ammonium Chloride Polar (soluble in water) Iodine Iodine Non polar (doesn’t dissolve in water) IS Copper (II) Sulfate IS Nathaline IS Copper (II) Sulfate IS Copper (II) Sulfate Non polar (doesn’t dissolve in water) Nathaline SS Nathaline Non polar (doesn’t dissolve in water) b. When a solute does not dissolve in water then it is considered to be polar, but if it doesn’t then it is considered to be nonpolar. 8. The rule ‘like dissolves like’ is useful when predicting solubility on most occasions. For example whether the substance is polar or nonpolar can decide whether or not the substance will be soluble or insoluble. 9 a. Water can be both a solute and a solvent. Water is considered to be a solvent when there are other elements being added to it. But on the other hand when water is the thing being added to something else it is a solute. b. With a substance you can usually tell whether it is a solute or a solvent because the solvent is usually the liquid which dissolves the solvent.