Chapter 4-Chemical Dominoes
advertisement

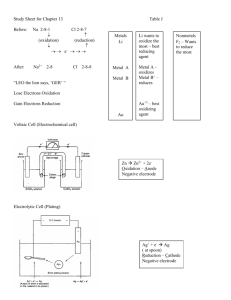
Chapter 4-Chemical Dominoes Section 6: Electrochemical Cells and Half-Reactions Pre-Test 1) Which of the following statements is true regarding solutions that conduct electricity? a. Solutions that conduct electricity contain neutral particles that do move around. b. Solutions that conduct electricity contain neutral particles that do not move around. c. Solutions that conduct electricity contain charged particles that do move around. d. Solutions that conduct electricity contain charged particles that do not move around. 2) When a metal loses electrons to form positive metal ions, this process is called oxidation. In an electrochemical cell, where does oxidation take place? a. In the cathode b. In the anode c. In both the anode and cathode d. In the salt bridge 3) When a metal loses electrons to form positive metal ions, this process is called oxidation. Which of the following balanced half-reactions represent the oxidation process? a. Zn Zn2+ + 2eb. Al3+ + 3e- Al c. 2 Ag+ + 2e- 2 Ag d. Na+1 + 1e- Na Use the activity series of metals below to answer question 4 and question 5. Activity Series of Metals Element lithium (most active) rubidium potassium barium calcium sodium magnesium aluminum manganese zinc chromium iron cobalt nickel tin lead *hydrogen copper mercury silver platinum gold (least active) Symbol Li Rb K Ba Ca Na Mg Al Mn Zn Cr Fe Co Ni Sn Pb H Cu Hg Ag Pt Au 4) When a metal loses electrons to form positive metal ions, this process is called oxidation. Which of the following metals would oxidize most easily? a. calcium b. chromium c. cobalt d. copper 5) Which of the following metals would be best to use as the cathode in an electrochemical cell? a. aluminum b. iron c. magnesium d. nickel