File
advertisement

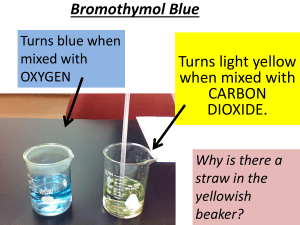
Name: _________________________ Station #3 CO2 Gas & Oxygen Deprivation—Silent Killer Introduction Carbon dioxide (CO2) is present in volcanic gas. It is invisible and odorless and is one of the major pollutants in the atmosphere, so it is critical we understand the hazards. Because CO 2 is heavier than air it can be deadly when concentrated in low lying areas. This inquiry-based demonstration shows how CO2 gas produced from vinegar and baking soda displaces oxygen and sequentially snuffs candles representing different elevations. The optional exercise uses dry ice to introduce change of matter from solid to gas as the ice sublimates, not passing through a liquid phase as H 2O ice does, under normal pressure conditions. Objectives • Learn about the toxicity of carbon dioxide; • See how carbon dioxide collects in low-lying areas; • Learn how to escape high carbon dioxide concentrations. Overview of Carbon Dioxide Gas Volcanic gases like carbon dioxide, sulfur dioxide (SO 2), hydrogen sulfide (H2S), and water (H2O), bubble out of magma as it rises to the surface. Volcanoes release more than 140 million tons of CO2 into the atmosphere every year. This colorless, odorless gas usually does not pose a direct hazard to life because it typically becomes diluted to low concentrations very quickly whether it is released continuously from the ground or during episodic eruptions. But in certain circumstances, CO2 may become concentrated at levels lethal to people and animals. Carbon dioxide gas is heavier than air and the gas can flow into in low-lying areas; breathing air with more than 30% CO2 can quickly induce unconsciousness and cause death. In volcanic or other areas where CO2 emissions occur, it is important to avoid small depressions and low areas that might be CO2 traps. The boundary between air and lethal gas can be extremely sharp; even a single step upslope may be adequate to escape death. There is normally between 300–500 ppm (0.03–0.05%) of CO2 in the atmosphere. Greater than this amount is considered pollution. The toxicity of CO2 is due to oxygen deprivation. At 2-3% in the air you might not notice it but just be short of breath. At 7% symptoms include headache, shortness of breath, dizziness, loss of mental ability, weakness, and ears ringing. At 10% you lose consciousness within 10-15 minutes. Above 15% is lethal. Again, the release of CO2 from volcanoes (from vents, the ground, and lava flows) is particularly hazardous because: (1) there is often little warning of high concentrations; (2) it is odorless and invisible; and (3) CO2 is heavier than air so it collects in topographically low and/or poorly ventilated places. Materials • Vinegar. • Baking soda. • 8- or 16-oz glass meas. cup with pouring lip (16 is best) • Soup bowl with room for three candles. • 3 candles (1/2, 1, and 11/2 inches high) • Optional Activity: dry ice (solid CO2). Name: _________________________ Station #3 Instructions 1. In your group talk about the role of CO2 in the atmosphere. Aside from exhaling, where else is CO2.produced? _____________________________________________________________________________________ _____________________________________________________________________________________ _____________________________________________________________________________________ 2. Place the two or three candles in the bottom of the bowl. Light the candles. 3. Pour 1/4 cup of vinegar into measuring cup. Cover all but the pour spout with plastic wrap. 4. Add a teaspoon of baking soda. Let it bubble for a bit. 5. Tip and pour spout over the bowl, careful not to let any bubbling mix spill out. Any gentle breeze will blow the gas away, so do this in a still room. Though invisible, the newly formed heavy gas will pour into the bowl, rising until first the shortest, then the tallest candles flicker, and then go out. 6. Record you observations and answer the following questions • What is going on? Detail your observations. ____________________________________________________________________________________ ____________________________________________________________________________________ ____________________________________________________________________________________ ____________________________________________________________________________________ ____________________________________________________________________________________ ____________________________________________________________________________________ ____________________________________________________________________________________ ____________________________________________________________________________________ ____________________________________________________________________________________ ____________________________________________________________________________________ ____________________________________________________________________________________ ____________________________________________________________________________________ • This gas is the same one which living things respire (breathe out)...so how can it be poisonous? ____________________________________________________________________________________ ____________________________________________________________________________________ ____________________________________________________________________________________ ____________________________________________________________________________________ ____________________________________________________________________________________ ____________________________________________________________________________________ Name: _________________________ Station #3 • What would happen if you were locked in a sealed room with excess vinegar and baking soda mixture? ____________________________________________________________________________________ ____________________________________________________________________________________ ____________________________________________________________________________________ ____________________________________________________________________________________ ____________________________________________________________________________________ ____________________________________________________________________________________ • What would you do if you knew that there was CO2 accumulation? ____________________________________________________________________________________ ____________________________________________________________________________________ ____________________________________________________________________________________ ____________________________________________________________________________________ ____________________________________________________________________________________ ____________________________________________________________________________________ • Where is the safest place in Victoria? ____________________________________________________________________________________ ____________________________________________________________________________________ 7. Read the Article “Volcanic Gas” and summarize the potential effects of the following volcanic gasses. Sulfur dioxide Formula ________ Potential effects: ____________________________________________________________________________________ ____________________________________________________________________________________ ____________________________________________________________________________________ ____________________________________________________________________________________ Hydrogen sulfide Formula ________ Potential effects: ____________________________________________________________________________________ ____________________________________________________________________________________ ____________________________________________________________________________________ ____________________________________________________________________________________ Name: _________________________ Station #3 Carbon dioxide Formula ________ Potential effects: ____________________________________________________________________________________ ____________________________________________________________________________________ ____________________________________________________________________________________ ____________________________________________________________________________________ Hydrogen chloride Formula ________ Potential effects: ____________________________________________________________________________________ ____________________________________________________________________________________ ____________________________________________________________________________________ ____________________________________________________________________________________ Hydrogen fluoride Formula ________ Potential effects: ____________________________________________________________________________________ ____________________________________________________________________________________ ____________________________________________________________________________________ ____________________________________________________________________________________